Chapter 18: Q. 38 (page 512)
of gas undergoes the process shown in Figure P16.38. What are
(a) temperature ,
(b) pressure , and
(c) volume ?
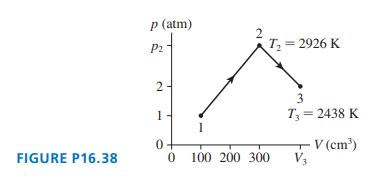
Short Answer
Part (a)
Part (b)
Part (c)
Learning Materials
Features
Discover
Chapter 18: Q. 38 (page 512)
of gas undergoes the process shown in Figure P16.38. What are
(a) temperature ,
(b) pressure , and
(c) volume ?

Part (a)
Part (b)
Part (c)
All the tools & learning materials you need for study success - in one app.
Get started for free
a. A sample of water vapour in an enclosed cylinder has an initial pressure of 500 Pa at an initial temperature of -0.01°C. A piston squeezes the sample smaller and smaller, without limit. Describe what happens to the water as the squeezing progresses.
b. Repeat part a if the initial temperature is 0.03°C warmer.
a. Is there a highest temperature at which ice can exist? If so,
what is it? If not, why not?
b. Is there a lowest temperature at which water vapor can exist?
If so, what is it? If not, why not?
of steam at a pressure of is cooled until it condenses. What is the volume of liquid water? Give your answer in .
An element in its solid phase has mass density 1750 kg/m3
and number density 4.3
A gas is in a Sealed container. The pressure is tripled and the temperature is doubled.
a. does the number of molecules within the container increase. decrease or stay the same?
b. By what factor does the number density of the gas increases.
What do you think about this solution?
We value your feedback to improve our textbook solutions.