Chapter 18: Q. 37 (page 512)
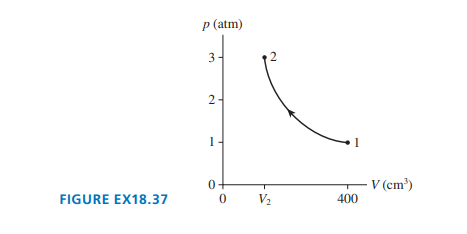
of gas undergoes the process shown in FIGURE EX18.37.
a. What type of process is this?
b. What is the final temperature in ?
c. What is the final volume ?
Short Answer
a. This type of process is called Isothermal.
b. The final temperature is ?
c. The final volume is?

