Chapter 18: Q. 2 (page 511)
The nucleus of a uranium atom has a diameter of 1.5 * 10-14 m
and a mass of 4.0 * 10-25 kg. What is the density of the nucleus?
Short Answer
The solution gives the description of the atoms of Uranium
Learning Materials
Features
Discover
Chapter 18: Q. 2 (page 511)
The nucleus of a uranium atom has a diameter of 1.5 * 10-14 m
and a mass of 4.0 * 10-25 kg. What is the density of the nucleus?
The solution gives the description of the atoms of Uranium
All the tools & learning materials you need for study success - in one app.
Get started for free
3.0 mol of gas at a temperature of -120C fills a 2.0 L
container. What is the gas pressure?
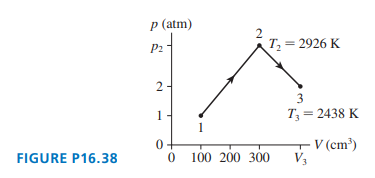
of gas undergoes the process shown in Figure P16.38. What are
(a) temperature ,
(b) pressure , and
(c) volume ?
The solar corona is a very hot atmosphere surrounding the visible surface of the sun. X-ray emissions from the corona show that its temperature is about . The gas pressure in the corona is about . Estimate the number density of particles in the solar corona.
A rigid container holds of gas at a pressure of and a temperature of .
a. What is the container’s volume?
b. What is the pressure if the temperature is raised to ?
What volume of water has the same mass as 100 cm3 of gold?
What do you think about this solution?
We value your feedback to improve our textbook solutions.