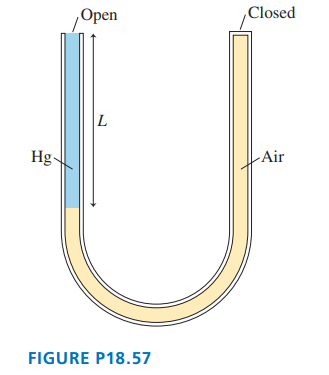
Chapter 18: Q. 4 (page 510)
The cylinder in FIGURE Q18.4 is divided into two compartments by a frictionless piston that can slide back and forth. If the piston is in equilibrium, is the pressure on the left side greater than, less than, or equal to the pressure on the right? Explain.
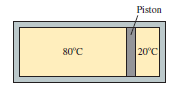
FIGURE Q18.4
Short Answer
As Force and Cross Sectional Area is same on either sides of the Piston, thus the pressure is same on Both Sides of the Piston from the two compartments.