Chapter 18: Q. 55 (page 513)
of steam at a pressure of is cooled until it condenses. What is the volume of liquid water? Give your answer in .
Short Answer
The volume of the liquid water is
Learning Materials
Features
Discover
Chapter 18: Q. 55 (page 513)
of steam at a pressure of is cooled until it condenses. What is the volume of liquid water? Give your answer in .
The volume of the liquid water is
All the tools & learning materials you need for study success - in one app.
Get started for free
The semiconductor industry manufactures integrated circuits in large vacuum chambers where the pressure is 1.0 * 10-10 mm of Hg.
a. What fraction is this of atmospheric pressure?
b. At T = 20C, how many molecules are in a cylindrical chamber 40 cm in diameter and 30 cm tall?
The solar corona is a very hot atmosphere surrounding the visible surface of the sun. X-ray emissions from the corona show that its temperature is about . The gas pressure in the corona is about . Estimate the number density of particles in the solar corona.
The atomic mass number of copper is A = 64. Assume that atoms in solid copper form a cubic crystal lattice. To envision this, imagine that you place atoms at the centers of tiny sugar cubes, then stack the little sugar cubes to form a big cube. If you dissolve the sugar, the atoms left behind are in a cubic crystal lattice. What is the smallest distance between two copper atoms?
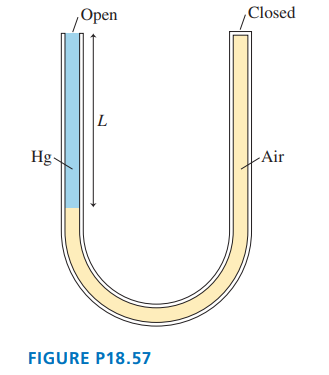
The U-shaped tube in FIGURE P18.57 has a total length of . It is open at one end, closed at the other, and is initially filled with air at pressure.
Mercury is poured slowly into the open end without letting any air escape, thus compressing the air. This is continued until the open side of the tube is completely filled with mercury. What is the length of the column of mercury?
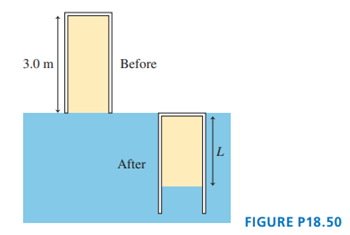
The -long pipe in FIGURE P18.50 is closed at the top end. It is slowly pushed straight down into the water until the top end of the pipe is level with the water’s surface. What is the length of the trapped volume of air?
What do you think about this solution?
We value your feedback to improve our textbook solutions.