Chapter 29: Q29-15P (page 1008)
What α-keto acid is formed on transamination of leucine?
Learning Materials
Features
Discover
Chapter 29: Q29-15P (page 1008)
What α-keto acid is formed on transamination of leucine?
All the tools & learning materials you need for study success - in one app.
Get started for free
PROBLEM 29-8Look at the entire glycolysis pathway, and make a list of the kinds of organic reactions that take place-nucleophilic acyl substitutions, aldol reactions, E1cB reactions, and so forth.
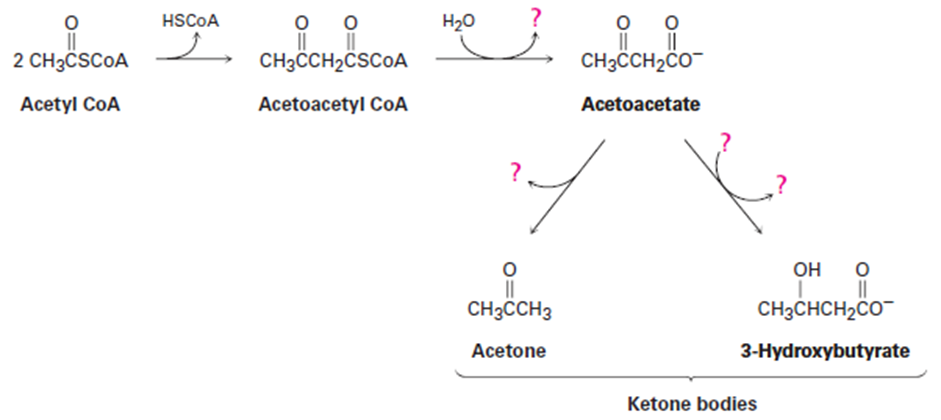
The primary fate of acetyl CoA under normal metabolic conditions is degradation in the citric acid cycle to yield CO2. When the body is stressed by prolonged starvation, however, acetyl CoA is converted into compounds called ketone bodies, which can be used by the brain as a temporary fuel. Fill in the missing information indicated by the four question marks in the following biochemical pathway for the synthesis of ketone bodies from acetyl CoA:
Give an mRNA sequence that will code for the synthesis of metenkephalin.
Tyr-Gly-Gly-Phe-Met
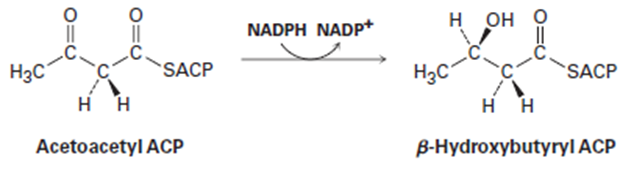
In step 6 of fatty-acid biosynthesis (Figure 29-5), acetoacetyl ACP is reduced stereospecifically by NADPH to yield an alcohol. Does hydride ion add to the Si face or the Re face of acetoacetyl ACP?
What general kind of reaction does NAD+ carry out?
What do you think about this solution?
We value your feedback to improve our textbook solutions.