Chapter 13: 54P (page 419)
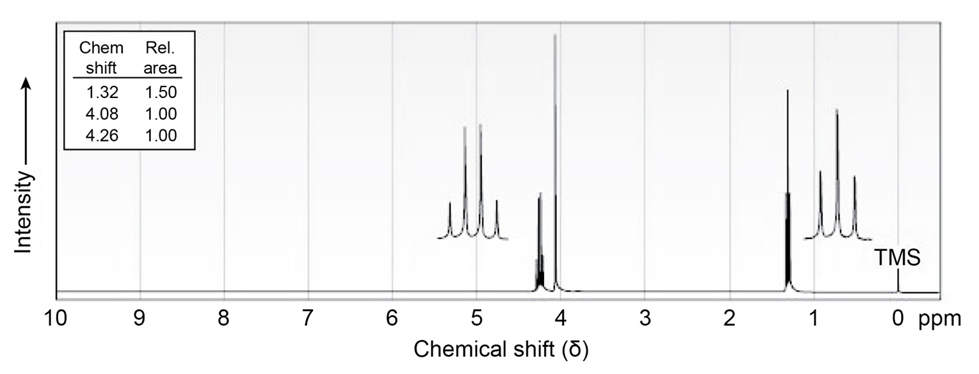
The compound whose 1H NMR spectrum is shown has the molecular
formula C4H7O2CI and has an infrared absorption peak at 1740cm-1.
Propose a structure.
Short Answer
Learning Materials
Features
Discover
Chapter 13: 54P (page 419)
The compound whose 1H NMR spectrum is shown has the molecular
formula C4H7O2CI and has an infrared absorption peak at 1740cm-1.
Propose a structure.


All the tools & learning materials you need for study success - in one app.
Get started for free
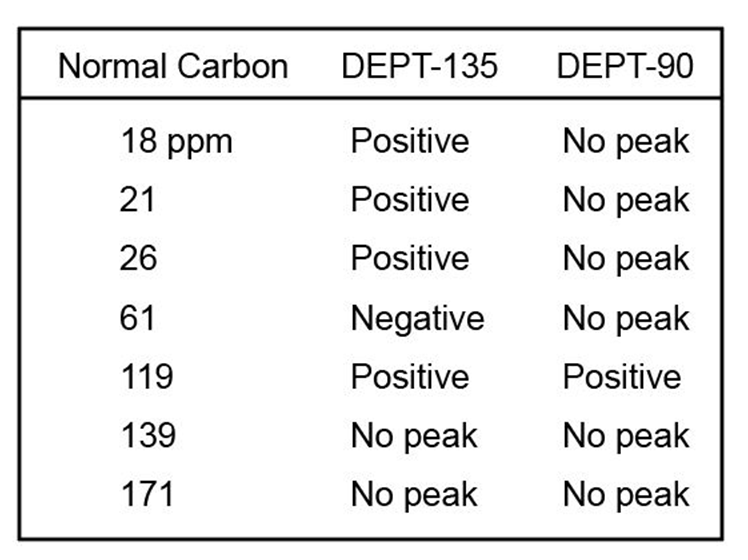
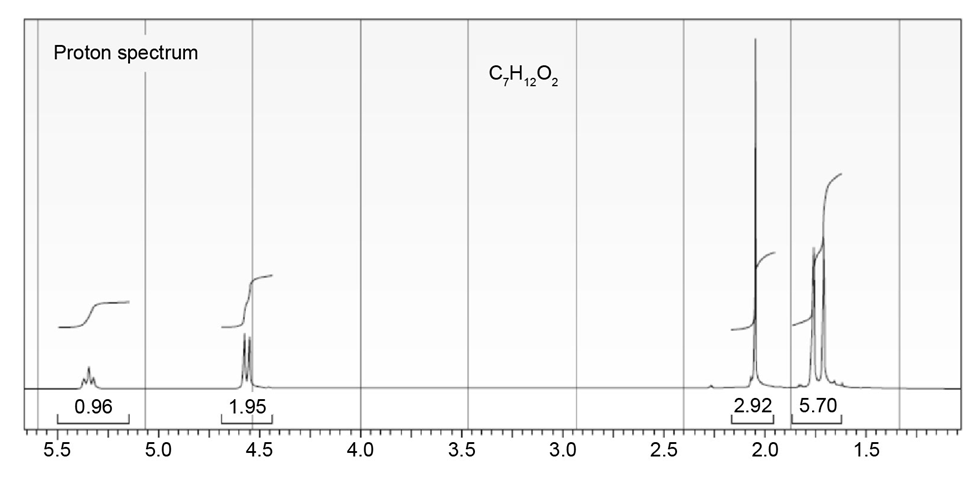
The proton NMR spectrum of a compound with the formula C7H12O2is
shown. The infrared spectrum displays a strong band at 1738 cm-1 and
a weak band at 1689 cm-1 . The normal carbon-13 and the DEPT experimental
results are tabulated. Draw the structure of this compound.
How could you use 1H NMR to determine the regiochemistry of electrophilic addition to alkenes? For example, does addition of HCl to 1-methylcyclohexene yield 1-chloro-1-methylcyclohexane or 1-chloro-2-methylcyclohexane?
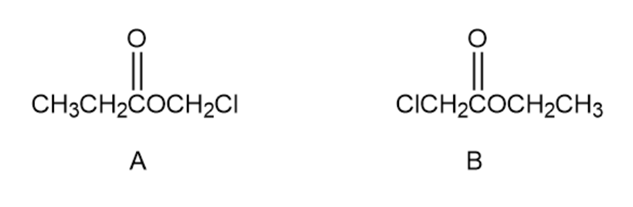
Question: The NMR spectra of compound A, , are shown. Propose a structure for A, and assign peaks in the spectra to your structure.
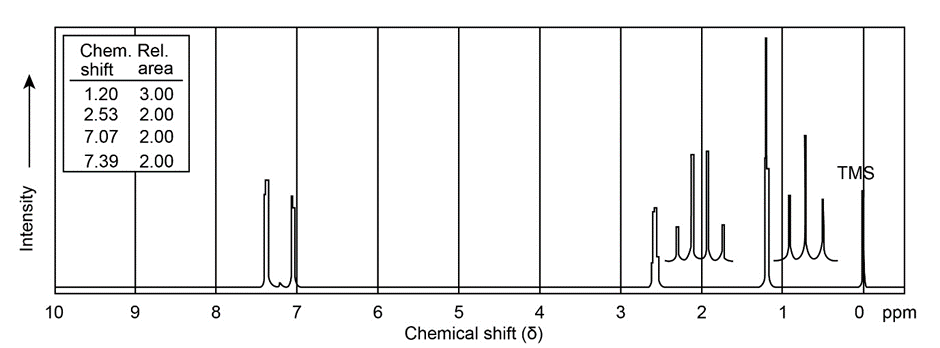
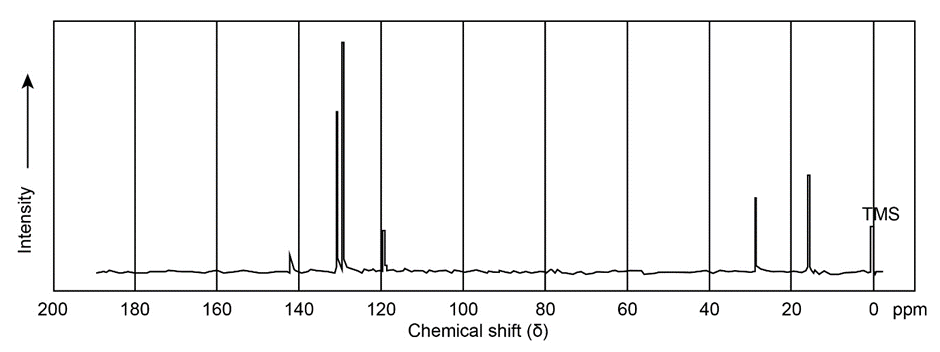
Propose a structure for compound E, , which has the following NMR spectral data:
Compound E Broadband-decoupled NMR: 19.1, 28.0, 70.5, 129.0, 129.8, 165.8 DEPT-90: 28.0, 129.8 DEPT-135: positive peaks at 19.1, 28.0, 129.8 ; negative peaks at 70.5, 129.0
Draw structures for compounds that meet the following descriptions:
(a) C2H6O ; one singlet
(b) C3H7CI ; one doublet and one septet
(c) C4H8CI2O ; two triplets
(d) C2H8O ; one singlet, one triplet, and one quartet
What do you think about this solution?
We value your feedback to improve our textbook solutions.