Chapter 13: 51P (page 419)
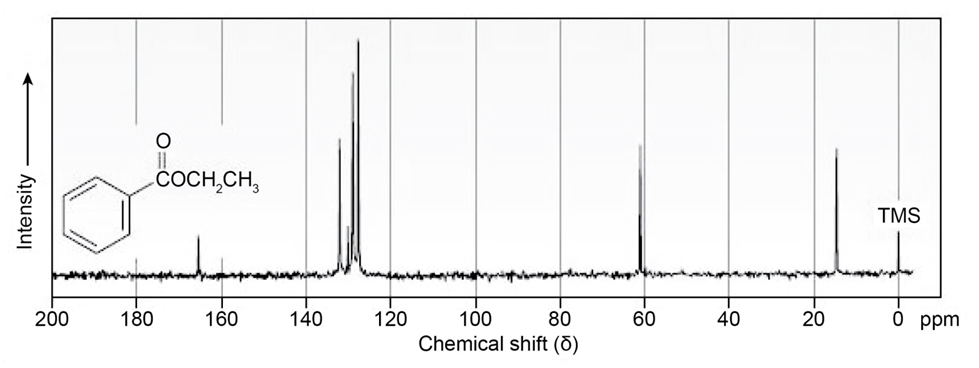
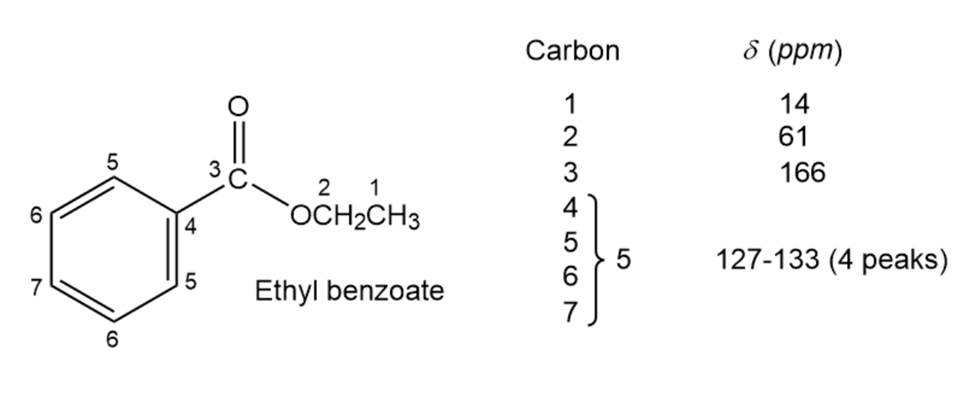
Assign as many resonances as you can to specific carbon atoms in the 13C
NMR spectrum of ethyl benzoate.
Short Answer
Learning Materials
Features
Discover
Chapter 13: 51P (page 419)
Assign as many resonances as you can to specific carbon atoms in the 13C
NMR spectrum of ethyl benzoate.


All the tools & learning materials you need for study success - in one app.
Get started for free
How many peaks would you expect in the 1H NMR spectrum of 1,4-dimethylbenzene(para-xylene, or p-xylene)? What ratio of peak areas would youexpect on integration of the spectrum? Refer to Table 13-3 for approximatechemical shifts, and sketch what the spectrum would look like. (Rememberfrom Section 2-4 that aromatic rings have two resonance forms.)
How many 13C NMR absorptions would you expect for cis-1,3-dimethylcyclohexane?For trans-1,3-dimethylcyclohexane? Explain.
The following 1H NMR absorptions were obtained on a spectrometer
operating at 300 MHz. Convert the chemical shifts from units to hertz
downfield from TMS.
(a)2.1(b)3.45(c)6.30(d)7.70
The compound whose 1H NMR spectrum is shown has the molecular
formula C4H7O2CI and has an infrared absorption peak at 1740cm-1.
Propose a structure.
Compound D is isomeric with compound C (Problem 13-61) and has the following NMR spectral data. Propose a structure.
Compound D Broadband-decoupled NMR: 9.7, 29.9, 74.4, 114.4, 141.4 d DEPT-90: 74.4, 141.4 d DEPT-135: positive peaks at 9.7, 74.4, 141.4 d; negative peaks at 29.9, 114.4 d
What do you think about this solution?
We value your feedback to improve our textbook solutions.