Chapter 12: Q59E (page 354)
Show how you might use a Wittig reaction to prepare the following alkenes. Identify the alkyl halide and the carbonyl components.
a.
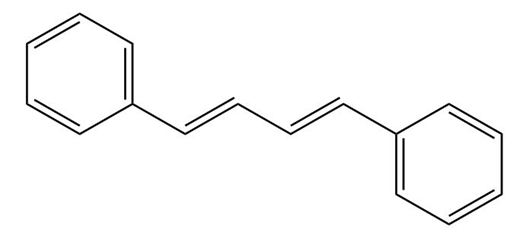
b.
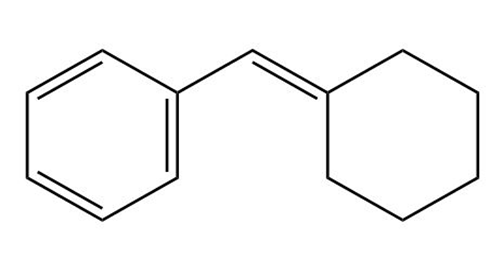
Short Answer
a.
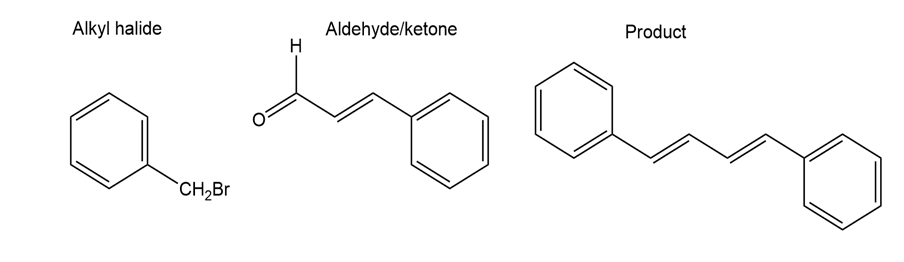
b.
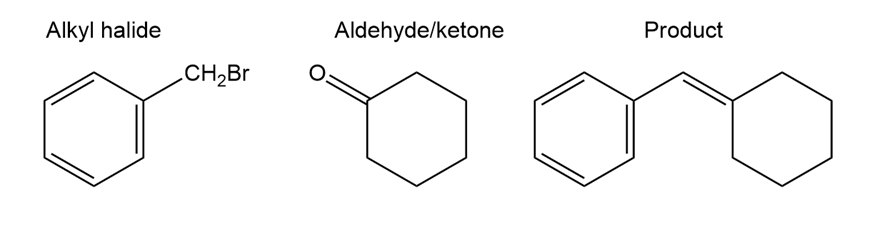
Learning Materials
Features
Discover
Chapter 12: Q59E (page 354)
Show how you might use a Wittig reaction to prepare the following alkenes. Identify the alkyl halide and the carbonyl components.
a.

b.

a.

b.

All the tools & learning materials you need for study success - in one app.
Get started for free
Question: At what approximate positions might the following compounds showIR absorptions?
(f)

Question: Camphor, a saturated monoketone from the Asian camphor tree, is used among other things as a moth repellent and as a constituent of embalming
fluid. If camphor has M+=152.1201 by high-resolution mass spectrometry,
what is its molecular formula? How many rings does camphor
have?
Question: How could you use infrared spectroscopy to distinguish between the
following pairs of isomers?
(a)
(b)
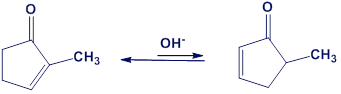
One consequence of the base-catalyzed isomerization of unsaturated ketones described in Problem 22-55 is that 2-substituted 2-cyclopentenones can be interconverted with 5-substituted 2-cyclopentenones. Propose a mechanism for this isomerization.
Assume that you are carrying out the dehydration of 1-methyl cyclohexanol to yield 1-methyl cyclohexene. How could you use infrared spectroscopy to determine when the reaction is complete?
What do you think about this solution?
We value your feedback to improve our textbook solutions.