Chapter 12: 13P (page 354)
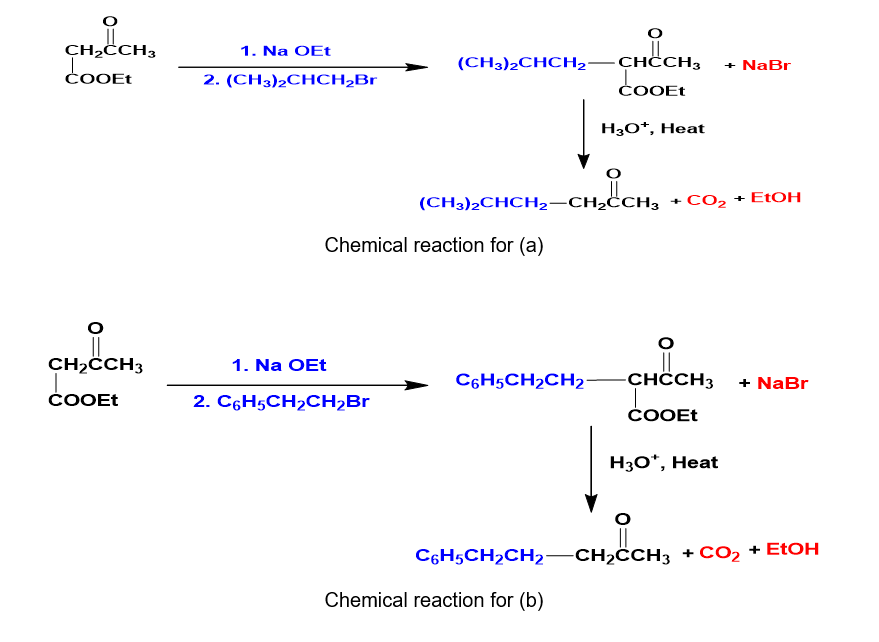
Question: What alkyl halides would you use to prepare the following ketones by an acetoacetic ester synthesis?
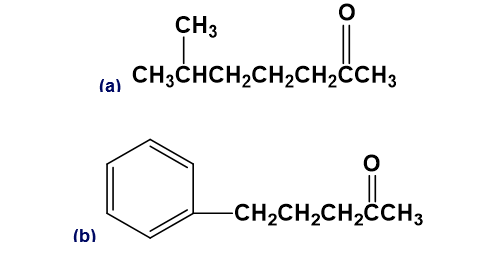
Short Answer
Learning Materials
Features
Discover
Chapter 12: 13P (page 354)
Question: What alkyl halides would you use to prepare the following ketones by an acetoacetic ester synthesis?


All the tools & learning materials you need for study success - in one app.
Get started for free
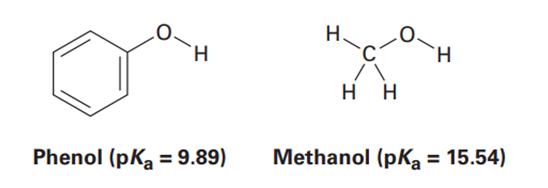
Phenol,, is a stronger acid than methanol, , even though both contain an OH bond. Draw the structures of the anions resulting from loss of from phenol and methanol, and use resonance structures to explain the difference in acidity.
Question: By knowing the natural abundances of minor isotopes, it’s possible to
calculate the relative heights of and M+1 peaks. If 13C has a natural
abundance of 1.10%, what are the relative heights of the and
M+1 peaks in the mass spectrum of benzene, C6H6?
Name the following alkynes, and predict the products of their reaction
With:
(1) in the presence of a Lindlar’s catalyst
(2) in the presence of
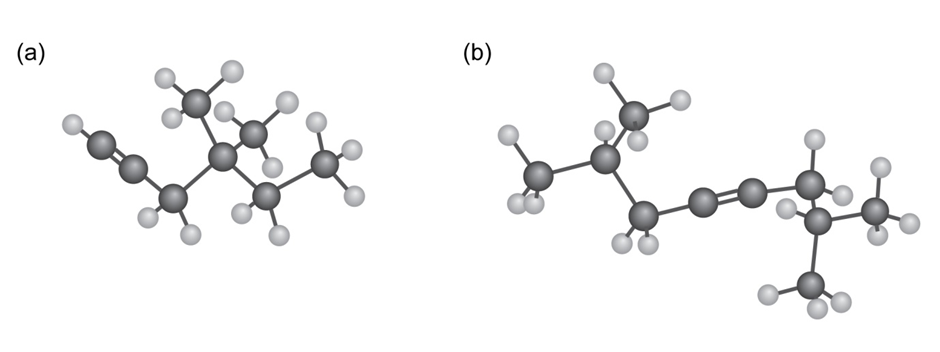
Rank the double bonds below in terms of increasing stability:
Question: How could you use infrared spectroscopy to distinguish between the
following pairs of isomers?
(a)
(b)
What do you think about this solution?
We value your feedback to improve our textbook solutions.