Chapter 1: Q1-15P (page 24)
Tell how many hydrogens are bonded to each carbon in the following compounds, and give the molecular formula of each substance:
a.
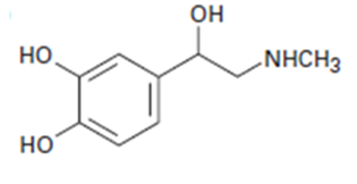
b.
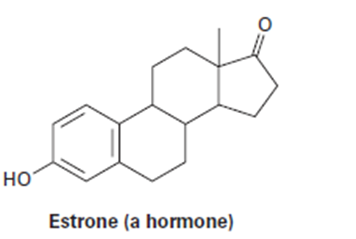
Short Answer
a.
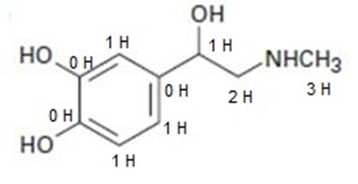
b.
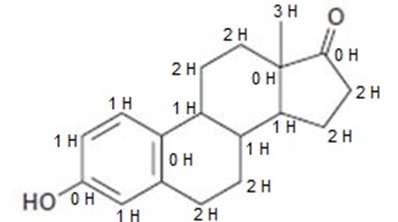
Learning Materials
Features
Discover
Chapter 1: Q1-15P (page 24)
Tell how many hydrogens are bonded to each carbon in the following compounds, and give the molecular formula of each substance:
a.

b.

a.

b.

All the tools & learning materials you need for study success - in one app.
Get started for free
Convert the following molecular model of hexane, a component of gasoline, into a line-bond structure (gray 5 C, ivory 5 H).
 HEXANE
HEXANE
Question: Draw a line-bond structure for 1, 3-butadiene . Indicate the hybridization of the orbitals on each carbon, and predict the value of each bond angle.
Oxaloacetic acid, an important intermediate in food metabolism, has the formulaand contains threebonds and twobonds. Propose two possible structures.
Draw a three-dimensional representation of the oxygen-bearing carbon atom in ethanol, using the standard convention of solid, wedged, and dashed lines.
the platelet aggregation inhibitor clopidogrel, marketed as Plavix. Give the molecular formula of each
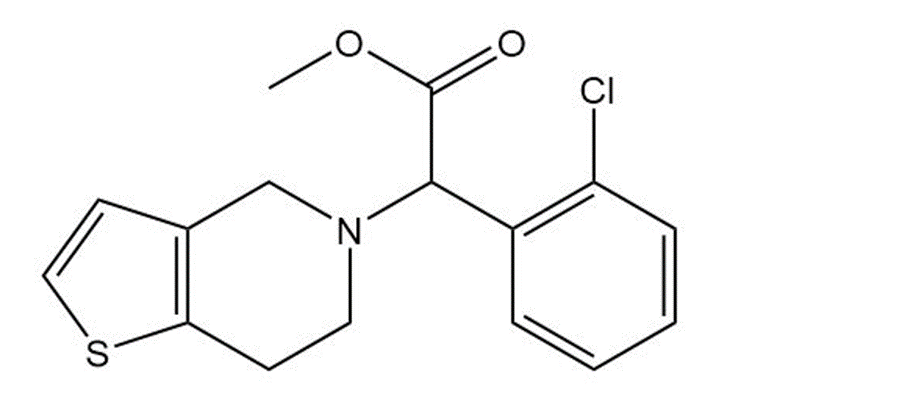
Clopidogrel (Plavix)
What do you think about this solution?
We value your feedback to improve our textbook solutions.