Chapter 5: Q52 E_a. (page 148)
Draw the meso form of each of the following molecules, and indicate the plane of symmetry in each:
(a)
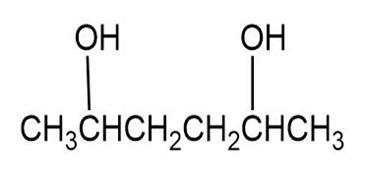
(b)
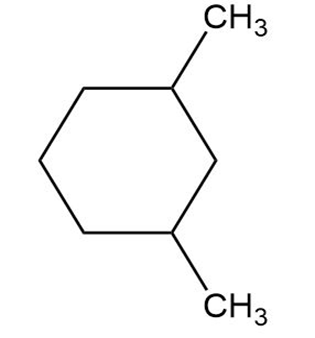
(c)
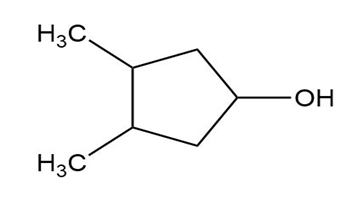
Short Answer
(a) The meso form of the given molecule is,
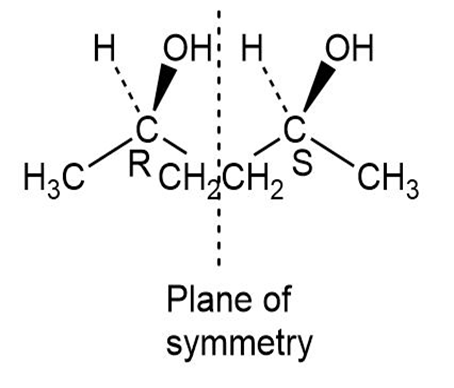
Learning Materials
Features
Discover
Chapter 5: Q52 E_a. (page 148)
Draw the meso form of each of the following molecules, and indicate the plane of symmetry in each:
(a)

(b)

(c)

(a) The meso form of the given molecule is,

All the tools & learning materials you need for study success - in one app.
Get started for free
One of the steps in fat metabolism is the hydration of crotonate to yield 3-hydroxybutyrate. This reaction occurs by addition of –OH to the Si face at C3, followed by protonation at C2, also from the Si face. Draw the product of the reaction, showing the stereochemistry of each step.
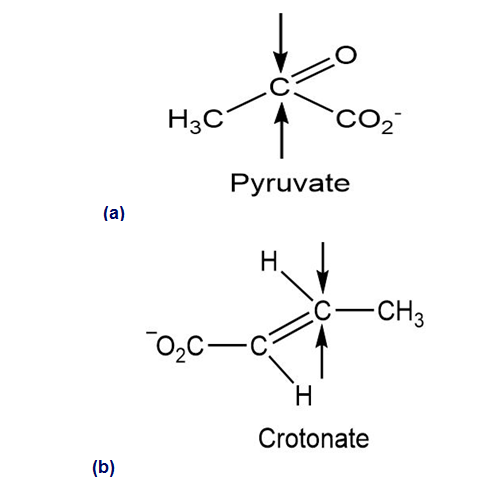
Identify the indicated faces in the following molecules as Re or Si:
Assign R or S stereochemistry to the chirality centers in the following
Newman projections:
Orient each of the following drawings so that the lowest-ranked group is toward the rear, and then assign R or S configuration
Which, if any, of the following structures represent meso compounds? (Blue = N, green = Cl.)
What do you think about this solution?
We value your feedback to improve our textbook solutions.