Chapter 5: Q. 30 E-a (page 148)
Orient each of the following drawings so that the lowest-ranked group is toward the rear, and then assign R or S configuration
Short Answer
(a). S configuration
Learning Materials
Features
Discover
Chapter 5: Q. 30 E-a (page 148)
Orient each of the following drawings so that the lowest-ranked group is toward the rear, and then assign R or S configuration
(a). S configuration
All the tools & learning materials you need for study success - in one app.
Get started for free
Question:Which of the following molecules are chiral? Identify the chirality center(s) in each.
(a)
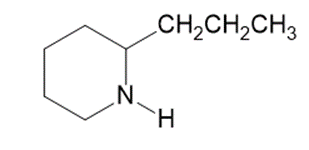
Coniine
(poison hemlock)
(b)
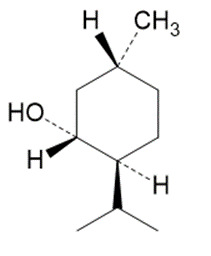
Menthol
(flavoring agent)
(c)
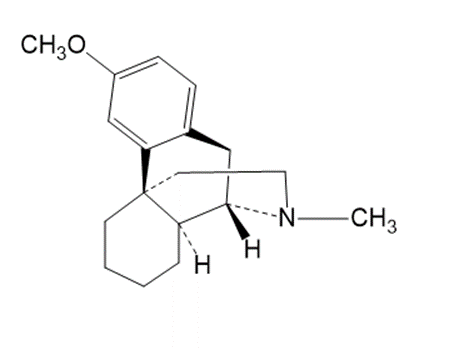
Dextromethorphan
(cough suppressant)
What is the stereochemical configuration of the enantiomer of (2S,4R)-2,4-octanediol? (A diol is a compound with two-OH groups.)
Identify the indicated hydrogens in the following molecules as pro-R or pro-S:
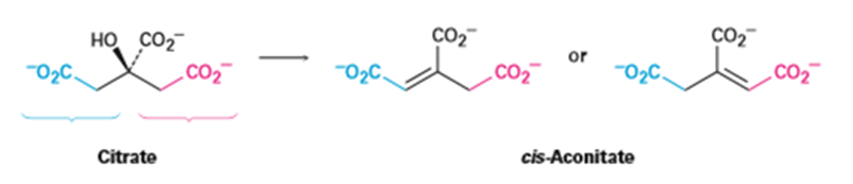
The dehydration of citrate to yield cis-aconitate, a step in the citric acid cycle, involves the pro-R “arm” of citrate rather than the pro-S arm. Which of the following two products is formed?
Question:Alanine, an amino acid found in proteins, is chiral. Draw the two enantiomers of alanine using the standard convention of solid, wedged, and dashed lines.
What do you think about this solution?
We value your feedback to improve our textbook solutions.