Chapter 5: Q. 56 E-c (page 148)
Identify the indicated hydrogens in the following molecules as pro-R or pro-S:
Learning Materials
Features
Discover
Chapter 5: Q. 56 E-c (page 148)
Identify the indicated hydrogens in the following molecules as pro-R or pro-S:
All the tools & learning materials you need for study success - in one app.
Get started for free
Orient each of the following drawings so that the lowest-ranked group is toward the rear, and then assign R or S configuration
A 1.50 g sample of coniine, the toxic extract of poison hemlock, was dissolved in 10.0 mL of ethanol and placed in a sample cell with a 5.00 cm pathlength. The observed rotation at the sodium D line was 11.21°. Calculate for coniine.
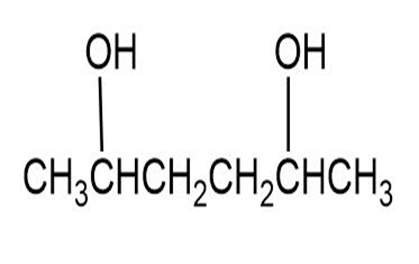
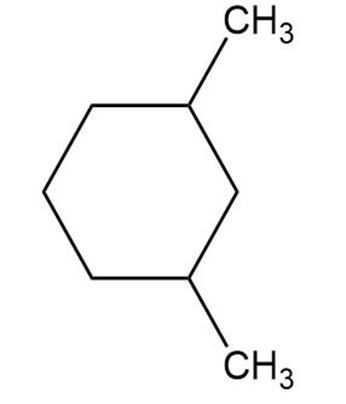
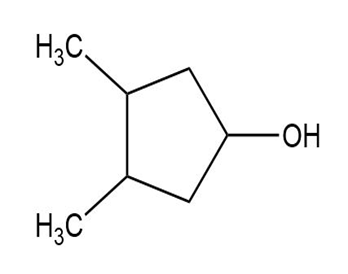
Draw the meso form of each of the following molecules, and indicate the plane of symmetry in each:
(a)
(b)
(c)
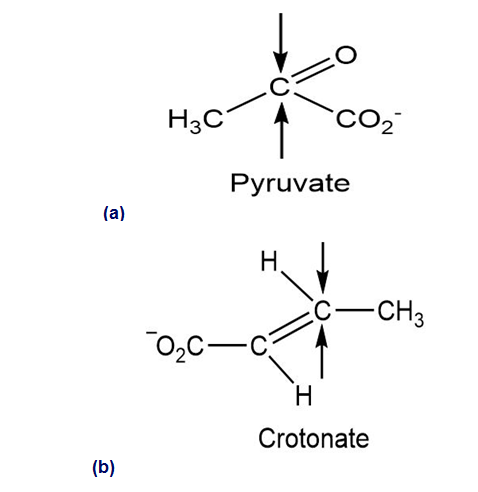
Identify the indicated faces in the following molecules as Re or Si:
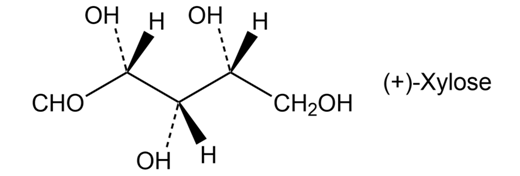
Xylose is a common sugar found in many types of wood, including maple and cherry. Because it is much less prone to cause tooth decay than sucrose, xylose has been used in candy and chewing gum. Assign R or S configurations to the chirality centers in xylose.
What do you think about this solution?
We value your feedback to improve our textbook solutions.