Chapter 5: Q. 30 E-c (page 148)
Orient each of the following drawings so that the lowest-ranked group is toward the rear, and then assign R or S configuration
Short Answer
(c). S configuration
Learning Materials
Features
Discover
Chapter 5: Q. 30 E-c (page 148)
Orient each of the following drawings so that the lowest-ranked group is toward the rear, and then assign R or S configuration
(c). S configuration
All the tools & learning materials you need for study success - in one app.
Get started for free
What is the relationship between the specific rotations of (2R,3R)-dichloropentane and (2S,3S)-dichloropentane? Between (2R,3S)-dichloropentane and (2R,3R)-dichloropentane?
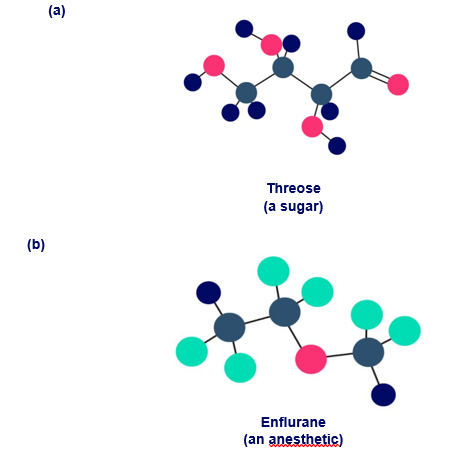
Question:Identify the chirality centers in the following molecules (green5Cl, yellow-green5F):
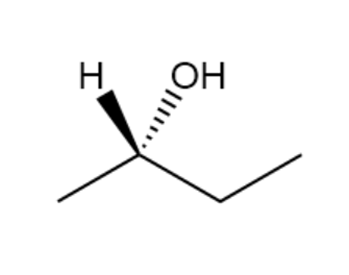
Assign R or S configurations to the chirality centers in the followingmolecules:
a.
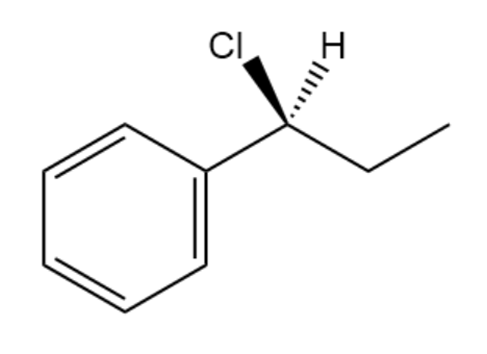
b.
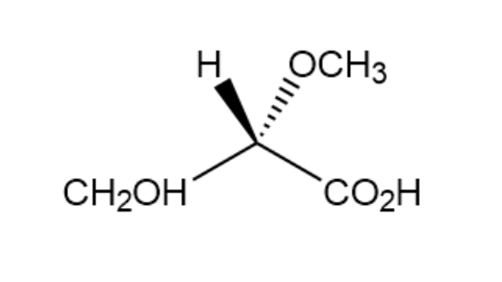
c.
Draw both cis- and trans-1,3-dimethyl cyclohexane in their more stable chair conformations.
(a) How many stereoisomers are there of cis-1,3-dimethyl cyclohexane, and how many of trans-1,3-dimethyl cyclohexane?
(b) Are any of the structures chiral?
(c) What are the stereochemical relationships among the various stereoisomers of 1,3-dimethyl cyclohexane?
Question:Which of the following objects are chiral?
(a) Soda can
(b)Screwdriver
(c)Screw
(d)Shoe
What do you think about this solution?
We value your feedback to improve our textbook solutions.