Chapter 5: Q. 27-a (page 148)
Assign R or S configurations to the chirality centers in the following molecules (blue = N)
Short Answer
- S- configuration
Learning Materials
Features
Discover
Chapter 5: Q. 27-a (page 148)
Assign R or S configurations to the chirality centers in the following molecules (blue = N)
All the tools & learning materials you need for study success - in one app.
Get started for free
Assign R or S configurations to the chirality centers in the followingmolecules:
a.
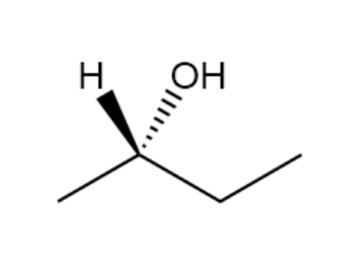
b.
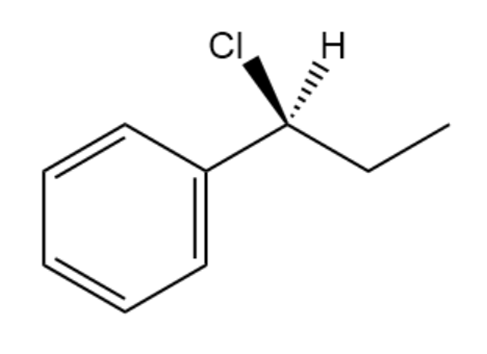
c.
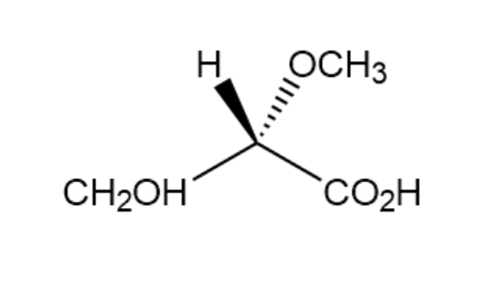
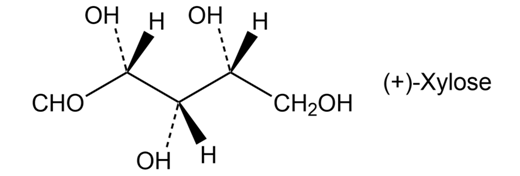
Xylose is a common sugar found in many types of wood, including maple and cherry. Because it is much less prone to cause tooth decay than sucrose, xylose has been used in candy and chewing gum. Assign R or S configurations to the chirality centers in xylose.
Question:Is cocaine (Worked Example 5-2) dextrorotatory or levorotatory?
Which, if any, of the following structures represent meso compounds? (Blue = N, green = Cl.)
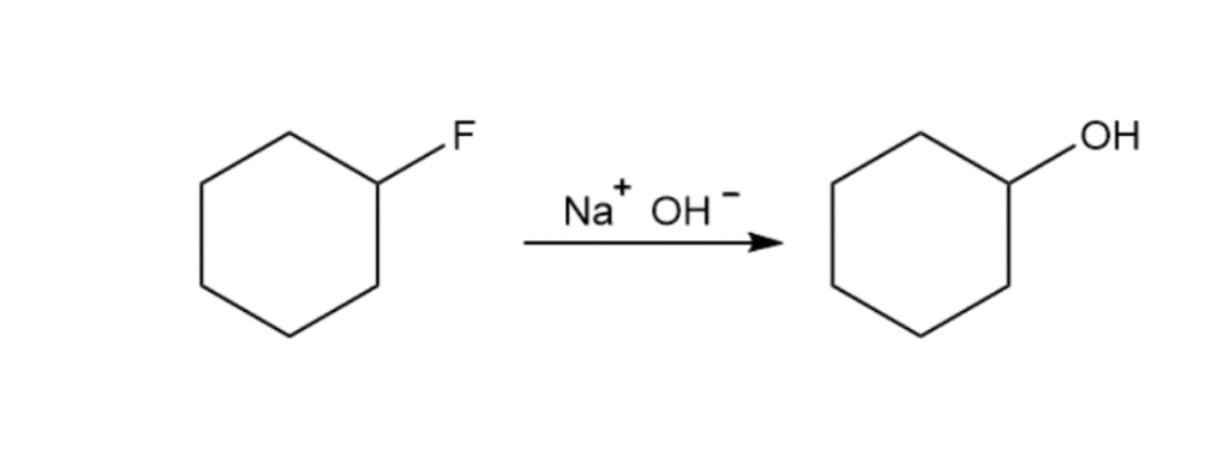
The reactions shown below are unlikely to occur as written. Tell what is wrong with each, and predict the actual product.
What do you think about this solution?
We value your feedback to improve our textbook solutions.