Chapter 5: 54bE (page 148)
The reactions shown below are unlikely to occur as written. Tell what is wrong with each, and predict the actual product.
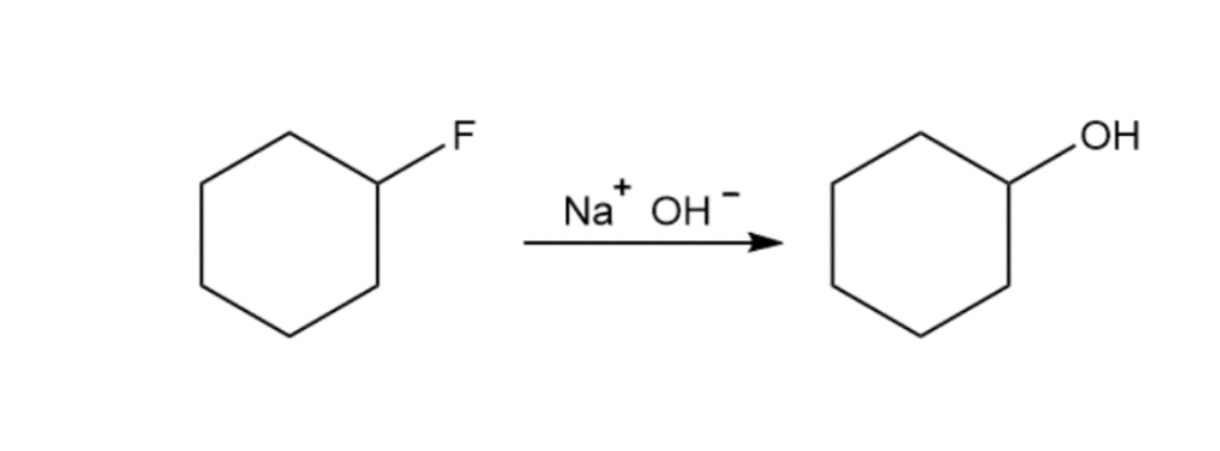
Short Answer
No, the reactions occurs beause the fluoride ion is a very poor leaving group.
Learning Materials
Features
Discover
Chapter 5: 54bE (page 148)
The reactions shown below are unlikely to occur as written. Tell what is wrong with each, and predict the actual product.

No, the reactions occurs beause the fluoride ion is a very poor leaving group.
All the tools & learning materials you need for study success - in one app.
Get started for free
What is the relationship between the specific rotations of (2R,3R)-dichloropentane and (2S,3S)-dichloropentane? Between (2R,3S)-dichloropentane and (2R,3R)-dichloropentane?
Question: Which member in each of the following sets ranks higher?
What is the stereochemical configuration of the enantiomer of (2S,4R)-2,4-octanediol? (A diol is a compound with two-OH groups.)
Which, if any, of the following structures represent meso compounds? (Blue = N, green = Cl.)
Draw tetrahedral representations of the following molecules: S-2-chlorobutane
What do you think about this solution?
We value your feedback to improve our textbook solutions.