Chapter 11: Q 11-11-42 E-d (page 350)
Which reactant in each of the following pairs is more nucleophilic? Explain.
d) (CH3)3P or (CH3)3N
Short Answer
(CH3)3P
Learning Materials
Features
Discover
Chapter 11: Q 11-11-42 E-d (page 350)
Which reactant in each of the following pairs is more nucleophilic? Explain.
d) (CH3)3P or (CH3)3N
(CH3)3P
All the tools & learning materials you need for study success - in one app.
Get started for free
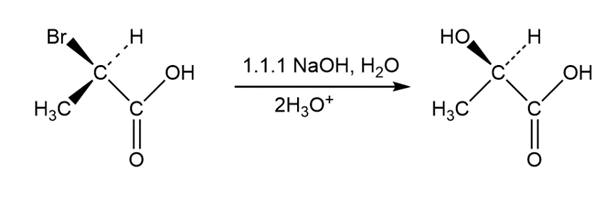
Question: Propose structures for compounds that fit the following descriptions:
(d) An alcohol that reacts rapidly with HCl at 0 °C
Draw the structure and assign Z or E stereochemistry to the product you expect from E2 reaction of the following molecule with NaOH (green = Cl):
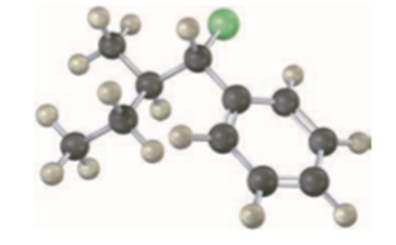
SN2 reactions take place with inversion of configuration, andSN1 reactions take place with racemization. The following substitution reaction, however, occurs with complete retention of configuration. Propose a mechanism.
.
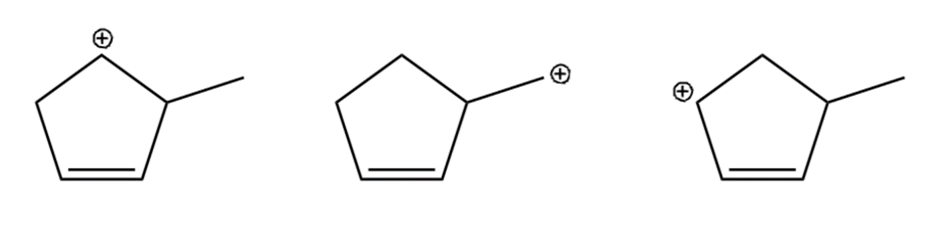
Question: Arrange the carbocations below in order of increasing stability.
a.
Reaction of iodoethane withyields a small amount of isonitrile,, along with the nitrileas the major product. Write electron-dot structures for both products, assign formal charges as necessary, and propose mechanisms to account for their formation.
What do you think about this solution?
We value your feedback to improve our textbook solutions.