Chapter 10: Q7P-a (page 297)
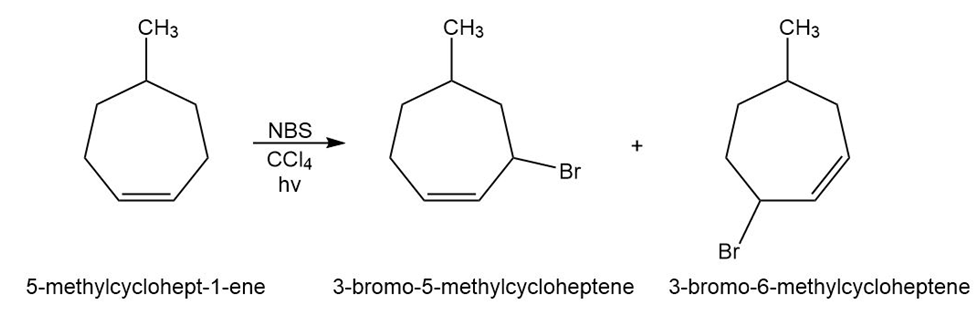
What products would you expect from reaction of the following alkenes with NBS? If more than one product is formed, show the structures of all.
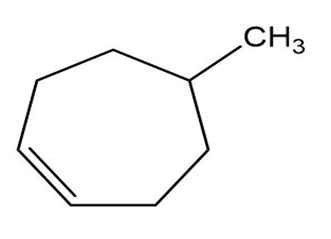
Short Answer
Learning Materials
Features
Discover
Chapter 10: Q7P-a (page 297)
What products would you expect from reaction of the following alkenes with NBS? If more than one product is formed, show the structures of all.


All the tools & learning materials you need for study success - in one app.
Get started for free
Draw structures corresponding to the following IUPAC names: (d) 1,1-Dibromo-4-isopropylcyclohexane.
Tell whether each of the following reactions is an oxidation, a reduction, or neither:
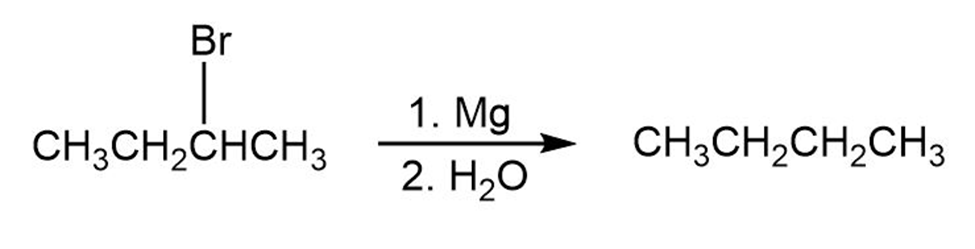
Draw and name all of the monochlorination products that you might obtain from the radical chlorination of the compounds below. Which of the products is chiral? Are any of the products optically active?
(a) 2-methylbutane
(b) Methylcyclopropane
(c) 2,2-dimethylpentane
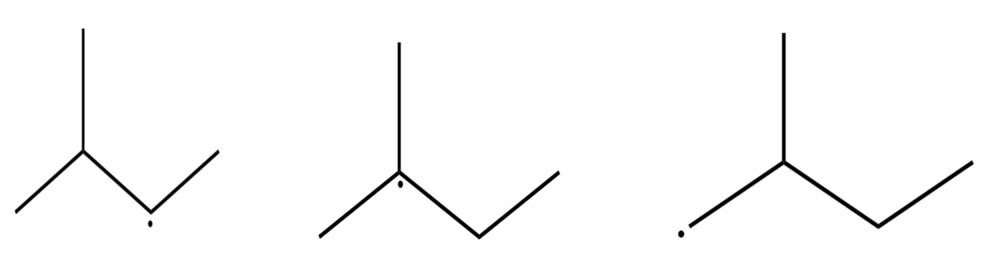
Draw three resonance forms for the cyclohexadienyl radical.
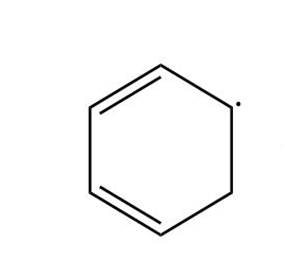
Cyclohexadienyl radical
Sort the radicals below from most stable to least stable.
What do you think about this solution?
We value your feedback to improve our textbook solutions.