Chapter 10: Q10-34E-c (page 308)
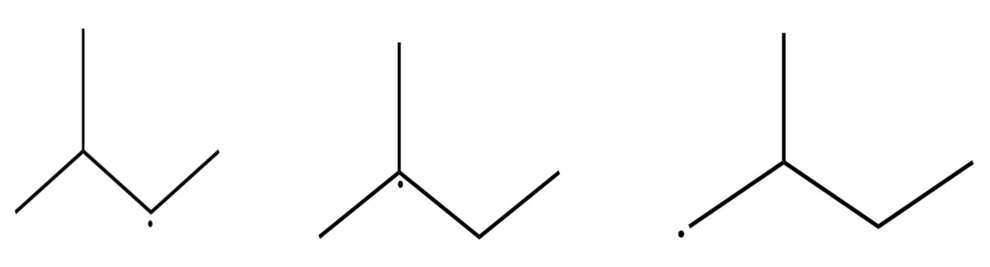
Sort the radicals below from most stable to least stable.
Short Answer
The order of radical stability is: 2 > 1 > 3
Learning Materials
Features
Discover
Chapter 10: Q10-34E-c (page 308)
Sort the radicals below from most stable to least stable.

The order of radical stability is: 2 > 1 > 3
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: (S)-3-Methylhexane undergoes radical bromination to yield optically inactive 3-bromo-3-methylhexane as the major product. Is the product chiral? What conclusions can you draw about the radical intermediate?
Alkyl halides can be reduced to alkanes by a radical reaction with tributyltinhydride, in the presence of light (hv). Propose aradical chain mechanism by which the reaction might occur. The initiationstep is the light-induced homolytic cleavage of the Sn-H bondto yield a tributyltin radical.
Carboxylic acids (RCOOH; =5) are approximately 1011 times more
acidic than alcohols (ROH;=16). In other words, a carboxylate ion
is more stable than an alkoxide ion . Explain, using
resonance.
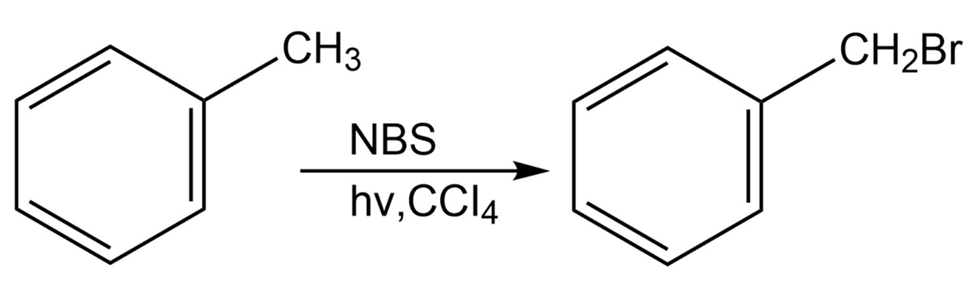
Alkylbenzenes such as toluene (methylbenzene) react with NBS to give
products in which bromine substitution has occurred next to the aromatic ring (the benzylic position). Explain, based on the bond dissociation energies in Table 6-3 on page 170.
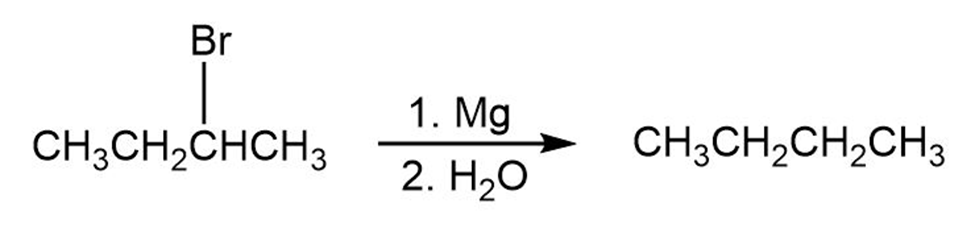
Tell whether each of the following reactions is an oxidation, a reduction, or neither:
What do you think about this solution?
We value your feedback to improve our textbook solutions.