Chapter 4: Q4-4-5 (page 89)
Draw the structures of the following molecules:
(a) trans-1-Bromo-3-methylcyclohexane
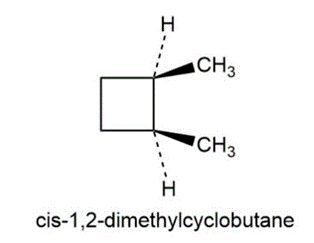
(b) cis-1,2-Dimethylcyclobutane
(c) trans-1-tert-Butyl-2-ethylcyclohexane
Short Answer
a)
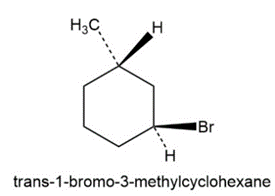
b)
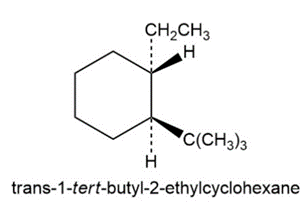
c)
Learning Materials
Features
Discover
Chapter 4: Q4-4-5 (page 89)
Draw the structures of the following molecules:
(a) trans-1-Bromo-3-methylcyclohexane
(b) cis-1,2-Dimethylcyclobutane
(c) trans-1-tert-Butyl-2-ethylcyclohexane
a)

b)

c)

All the tools & learning materials you need for study success - in one app.
Get started for free
In light of your answer to Problem 4-48, draw the two chair conformations of 1,1,3-trimethylcyclohexane and estimate the amount of strain energy in each. Which conformation is favored?
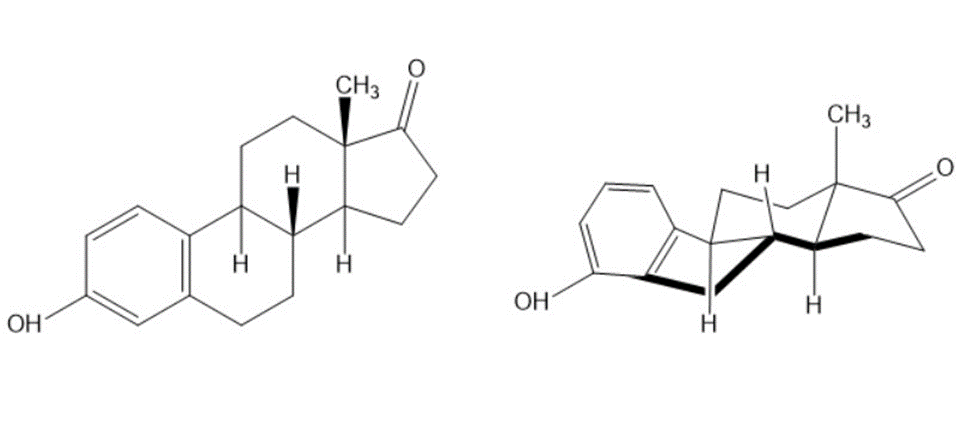
Look at the following structure of the female hormone estrone, and tell whether each of the two indicated ring-fusions is cis or trans.
How many eclipsing interactions would be present if cyclopentanewere planar? Assuming an energy cost of 4.0 kJ/mol for each eclipsing interaction,how much torsional strain would planar cyclopentane have? Since themeasured total strain of cyclopentane is 26 kJ/mol, how much of the torsionalstrain is relieved by puckering?
Two conformations of cis-1,3-dimethylcyclobutane are shown. What is the difference between them, and which do you think is likely to be more stable?

Why do you suppose an axial cyano (–CN) substituent causes practically no 1,3-diaxial steric strain (0.4 kJ/mol)? Use molecular models to help with your answer.
What do you think about this solution?
We value your feedback to improve our textbook solutions.