Chapter 3: Q39E (page 88)
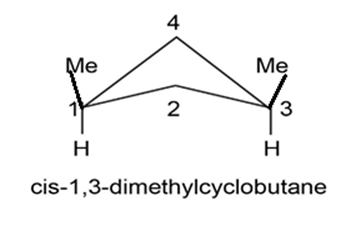
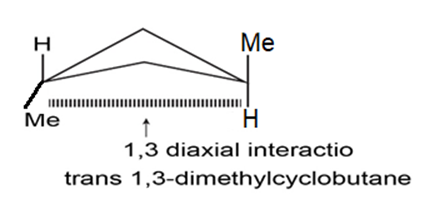
cis-1, 2-Dimethylcyclobutane is less stable than its trans isomer, but cis-1, 3-dimethylcyclobutane is more stable than its trans isomer. Draw the most stable conformations of both, and explain.
Short Answer
Cis-1, 2-Dimethylcyclobutane is less stable than its trans isomer because of eclipsed strain observed in cis-1, 2-dimethylcyclobutane.
 Cis-1,2-dimethylcyclobutane
Cis-1,2-dimethylcyclobutane Trans-1,2-dimethylcyclobutane
Trans-1,2-dimethylcyclobutane