Chapter 3: Q11 P-a (page 78)
Give IUPAC names for the following compounds:
(a)The three isomers of C5H12
Short Answer
(a)The IUPAC name of three isomers of C5H12are pentane, 2-Methylbutane and 2,2-Dimethylpropane.
Learning Materials
Features
Discover
Chapter 3: Q11 P-a (page 78)
Give IUPAC names for the following compounds:
(a)The three isomers of C5H12
(a)The IUPAC name of three isomers of C5H12are pentane, 2-Methylbutane and 2,2-Dimethylpropane.
All the tools & learning materials you need for study success - in one app.
Get started for free
Give IUPAC names for the following compounds:
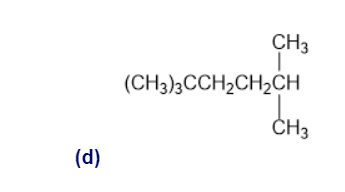
Draw and name all monobromo derivatives of pentane .
Identify the carbon atoms in the following molecules as primary, secondary, tertiary, or quaternary
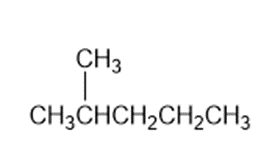
a)
Increased substitution around a bond leads to increased strain. Take the four substituted butanes listed below, for example. For each compound, sight along the C2–C3 bond and draw Newman projections of the most stable and least stable conformations. Use the data in Table 3-5 to assign strain-energy values to each conformation. Which of the eight conformations is most strained? Which is least strained?
(a) 2-Methylbutane
(b) 2,2-Dimethylbutane
(c) 2,3-Dimethylbutane
(d) 2,2,3-Trimethylbutane
Locate and identify the functional groups in the following molecules.
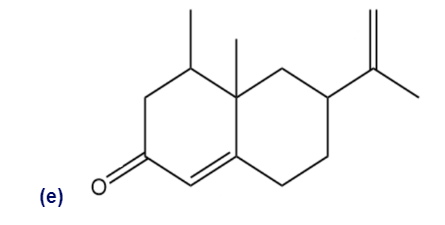
What do you think about this solution?
We value your feedback to improve our textbook solutions.