Chapter 30: Q39E (page 1033)
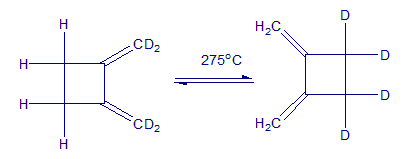
The following reaction is an example of a [2,3] sigmatropic rearrangement. Would you expect the reaction to be suprafacial or antarafacial? Explain.
Short Answer
A [2,3] sigmatropic rearrangement involves three electron pairs (one π bond, one lone pair, and one σ bond), the orbital-symmetry rules predict a suprafacial reaction