Chapter 30: Orbitals and Organic Chemistry: Pericyclic Reactions
Q.30-18
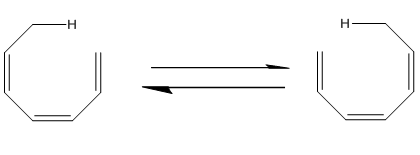
Do the following electrocyclic reactions take place in a conrotatory or
disrotatory manner? Under what conditions, thermal or photochemical, would you carry out each reaction?
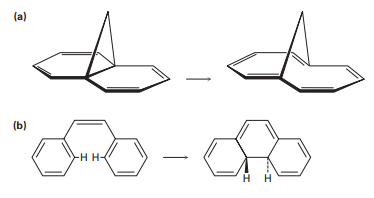
Q.30-19
The following thermal isomerization occurs under relatively mild conditions. Identify the pericyclic reactions involved, and show how the rearrangement occurs.
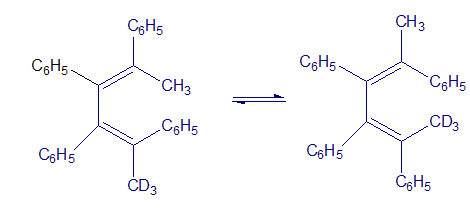
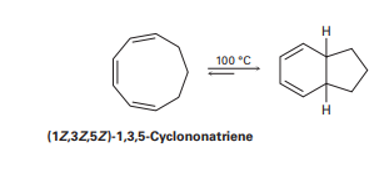
Q.30-21E
Heating (1Z,3Z,5Z)-1,3,5-cyclononatriene to 100°C causes cyclization and formation of a bicyclic product. Is the reaction conrotatory or disrotatory? What is the stereochemical relationship of the two hydrogens at the ring junctions, cis or trans?
Q.30-22E
(2E,4Z,6Z,8E)-2,4,6,8-Decatetraene has been cyclized to give 7,8-dimethyl-1,3,5-cyclooctatriene. Predict the manner of ring-closure- conrotatory or disrotatory for both thermal and photochemical reactions, and predict the stereochemistry of the product in each case.
Q.30-23E
Answer Problem 30-22 for the thermal and photochemical cyclizations of (2E,4Z,6Z,8Z)-2,4,6,8-decatetraene.
Q.30-3
trans-3,4-Dimethylcyclobutene can open by two conrotatory paths to give either (2E,4E)-2,4-hexadiene or (2Z,4Z)-2,4-hexadiene. Explain why both products are symmetry-allowed, and then account for the fact that only the 2E,4Eisomer is obtained in practice.
Q.30-4
What product would you expect to obtain from the photochemical cyclizationof (2E,4Z,6E)-2,4,6-octatriene? Of (2E,4Z,6Z)-2,4,6-octatriene?
Q.30-5
What stereochemistry would you expect for the product of the Diels–Alder reaction between (2E,4E)-2,4-hexadiene and ethylene? What stereochemistry would you expect if (2E,4Z)-2,4-hexadiene were used instead?
Q.30-6
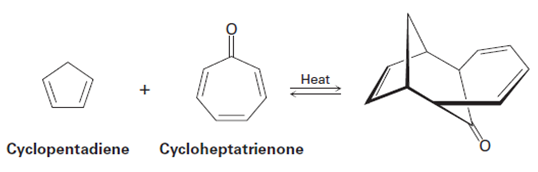
1,3-Cyclopentadiene reacts with cycloheptatrienone to give the productshown. Tell what kind of reaction is involved, and explain the observed result.Is the reaction suprafacial or antarafacial?
Q.30-7
Classify the following sigmatropic reaction by order [x,y], and tell whether itwill proceed with suprafacial or antarafacial stereochemistry: