Chapter 30: Q.30-19 (page 1033)
The following thermal isomerization occurs under relatively mild conditions. Identify the pericyclic reactions involved, and show how the rearrangement occurs.
Short Answer
The Conversion can be shown as
Learning Materials
Features
Discover
Chapter 30: Q.30-19 (page 1033)
The following thermal isomerization occurs under relatively mild conditions. Identify the pericyclic reactions involved, and show how the rearrangement occurs.

The Conversion can be shown as

All the tools & learning materials you need for study success - in one app.
Get started for free
trans-3,4-Dimethylcyclobutene can open by two conrotatory paths to give either (2E,4E)-2,4-hexadiene or (2Z,4Z)-2,4-hexadiene. Explain why both products are symmetry-allowed, and then account for the fact that only the 2E,4Eisomer is obtained in practice.
What stereochemistry would you expect for the product of the Diels–Alder reaction between (2E,4E)-2,4-hexadiene and ethylene? What stereochemistry would you expect if (2E,4Z)-2,4-hexadiene were used instead?
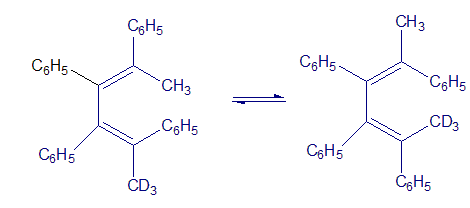
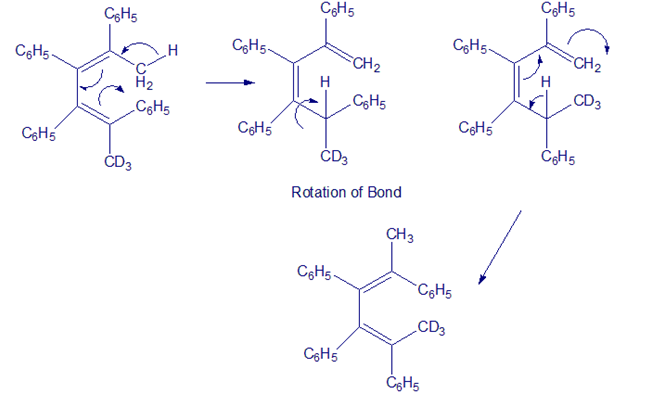
The following rearrangement was devised and carried out to prove the stereochemistry of [1,5] sigmatropic hydrogen shifts. Explain how the observed result confirms the predictions of orbital symmetry.
reaction involved.
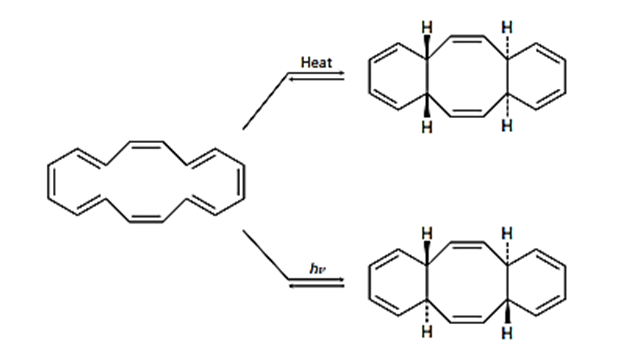
The cyclohexadecaoctaene shown isomerizes to two different isomers, depending on reaction conditions. Explain the observed results, and indicate whether each reaction is conrotatory or disrotatory.
What do you think about this solution?
We value your feedback to improve our textbook solutions.