Chapter 18: Q70E (page 594)
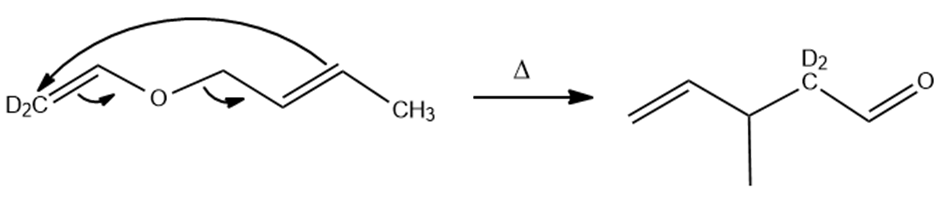
Predict the product(s) if the starting materials below underwent a Claisen rearrangement. Draw arrows to illustrate the rearrangement of electrons.
(a)
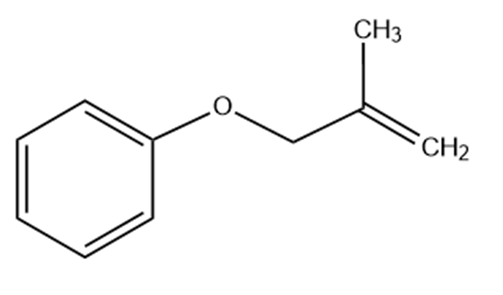
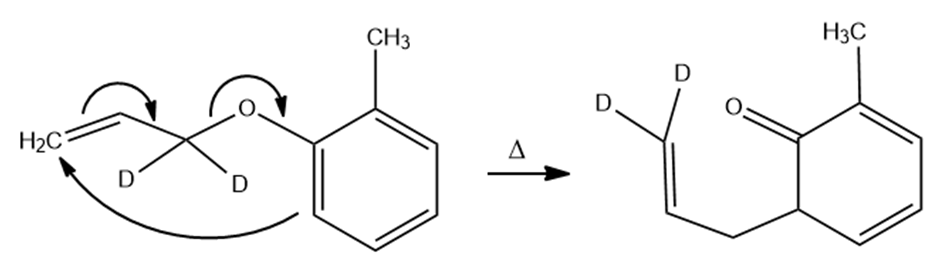
(b)
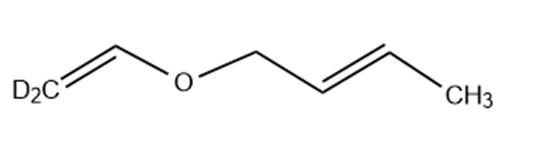
(c)
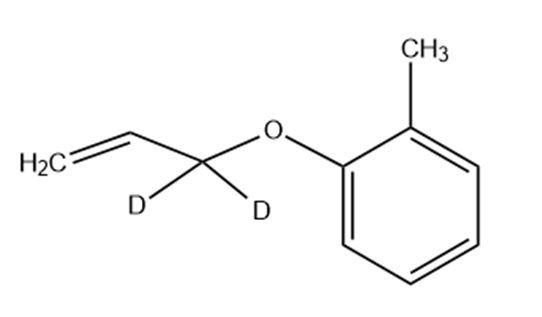
Short Answer
The answer is
(a)
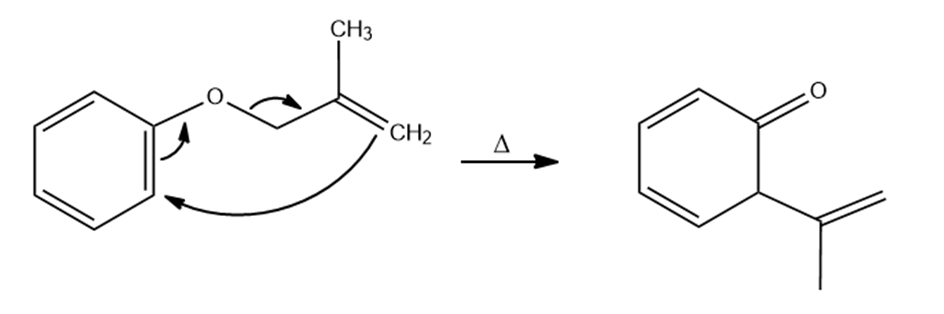
(b)
(c)
Learning Materials
Features
Discover
Chapter 18: Q70E (page 594)
Predict the product(s) if the starting materials below underwent a Claisen rearrangement. Draw arrows to illustrate the rearrangement of electrons.
(a)

(b)

(c)

The answer is
(a)

(b)

(c)

All the tools & learning materials you need for study success - in one app.
Get started for free
Treatment of the following alkene with a peroxy acid yields an epoxide different from that obtained by reaction with aqueous Br2 followed by base treatment. Propose structures for the two epoxides, and explain the result.
Show the product, including stereochemistry, that would result from reaction of the following epoxide with HBr
Aldehydes and ketones undergo acid-catalyzed reaction with alcoholsto yield hemiacetals,compounds that have one alcohol-like oxygenand one ether-like oxygen bonded to the same carbon. Further reactionof a hemiacetal with alcohol then yields an acetal,a compound that hastwo ether-like oxygens bonded to the same carbon.
(a)Show the structures of the hemiacetal and acetal you would obtainby reaction of cyclohexanone with ethanol.
(b)Propose a mechanism for the conversion of a hemiacetal into anacetal.
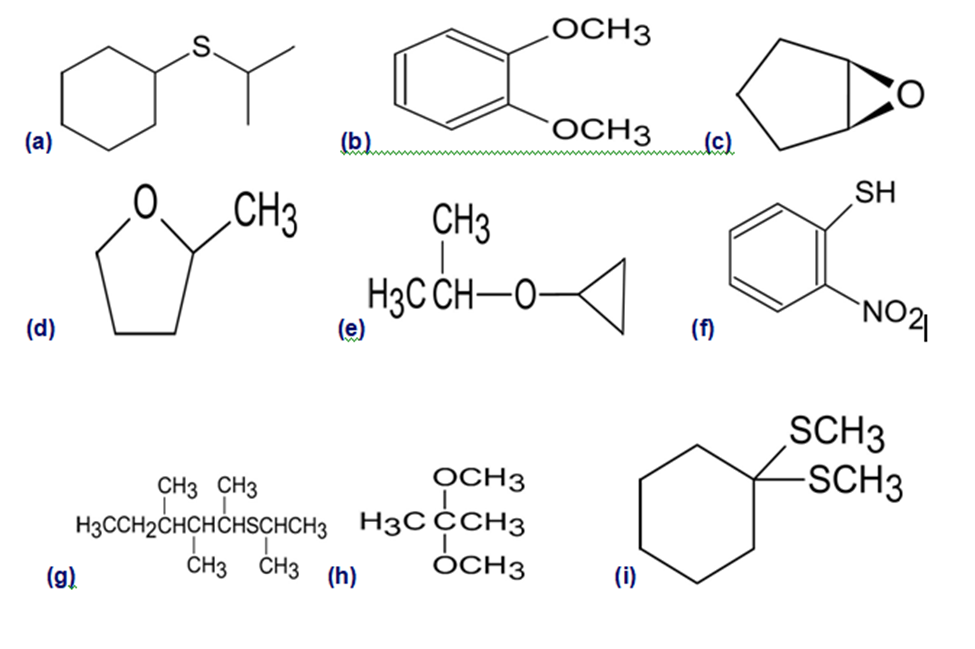
Give IUPAC names for the following structures:
Griginard reagents react with oxetane, a four membered cyclic ether, to yield primary alcohols, but the reaction is much slower than the corresponding reaction with ethylene oxide. Suggest a reason for the difference in reactivity between oxetane and ethylene oxide.
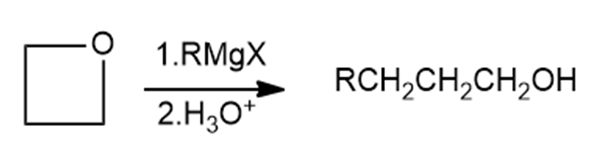
Oxetane
What do you think about this solution?
We value your feedback to improve our textbook solutions.