Chapter 18: Q56E (page 594)
Predict the products of the following reactions:
Short Answer
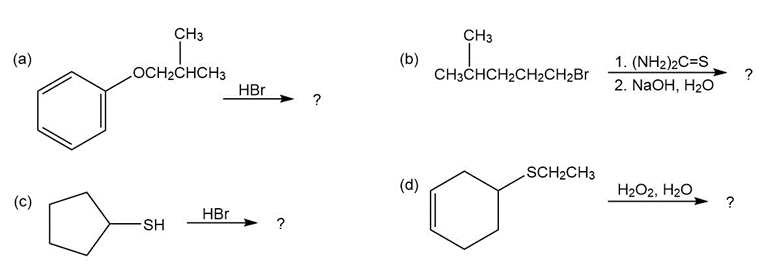
a)
(Products of reaction (a)
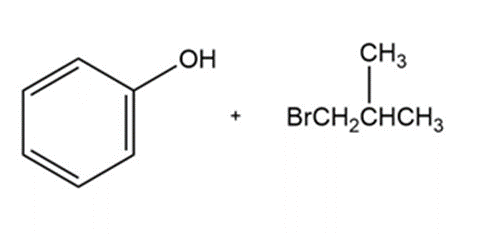
b)
Product of reaction (b)
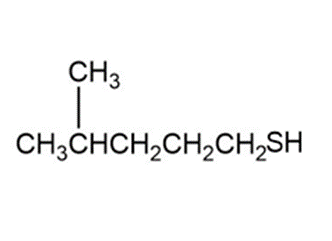
c)
Product of reaction (c)
d)
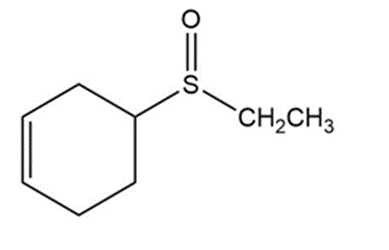
Product of reaction (d)
Learning Materials
Features
Discover
Chapter 18: Q56E (page 594)
Predict the products of the following reactions:

a)

(Products of reaction (a)
b)

Product of reaction (b)
c)

Product of reaction (c)
d)

Product of reaction (d)
All the tools & learning materials you need for study success - in one app.
Get started for free
How could you prepare benzyl phenyl ether from benzene and phenol? More than one step is required.
Why do you suppose only symmetrical ethers are prepared by the sulfuric acid-catalyzed dehydration procedure? What product (s) would you expect if ethanol and 1-propanol were allowed to react together? In what ratio would the products be formed if the two alcohols were of equal reactivity?
Review the mechanism of oxymercuration shown in Figure 8-3 on page 230, and then write the mechanism of the alkoxymercuration reaction of 1-methyl-cyclopentene with ethanol. Use curved arrows to show the electron flow in each step.
Predict the product(s) if the starting materials below underwent a Claisen rearrangement. Draw arrows to illustrate the rearrangement of electrons.
(a)
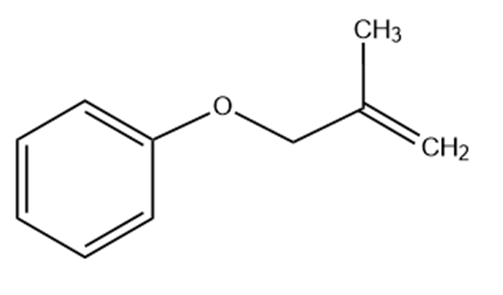
(b)
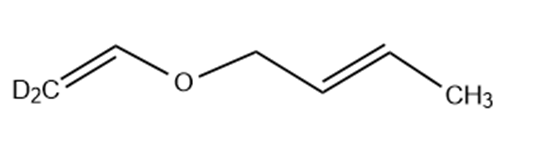
(c)
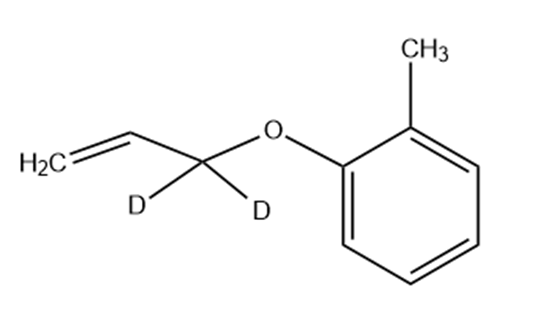
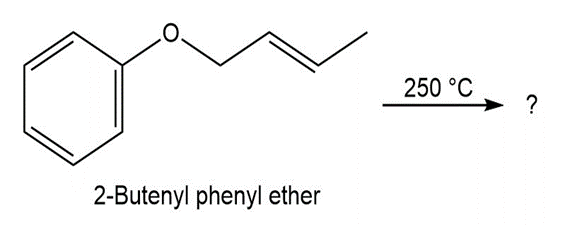
What product would you expect from Claisen rearrangement of 2-butenyl phenyl ether?
What do you think about this solution?
We value your feedback to improve our textbook solutions.