Chapter 18: 18-18-8P (page 575)
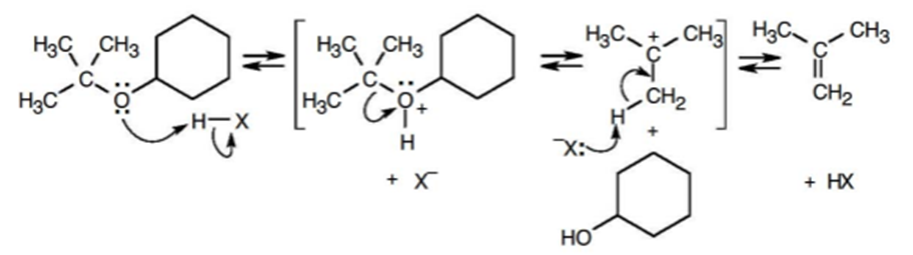
Write the mechanism of the acid-induced cleavage of tert-butyl cyclohexyl ether to yield cyclohexanol and 2-methylpropene.
Short Answer
The mechanism of the acid-induced cleavage of tert-butyl cyclohexyl ether is,
Learning Materials
Features
Discover
Chapter 18: 18-18-8P (page 575)
Write the mechanism of the acid-induced cleavage of tert-butyl cyclohexyl ether to yield cyclohexanol and 2-methylpropene.
The mechanism of the acid-induced cleavage of tert-butyl cyclohexyl ether is,

All the tools & learning materials you need for study success - in one app.
Get started for free
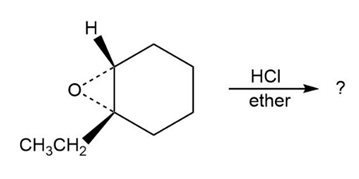
Predict the product(s) and provide the mechanism for each reaction below. What do the mechanisms have in common?
a.
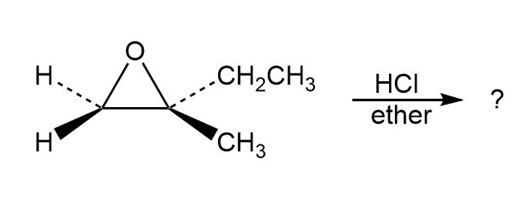
b.
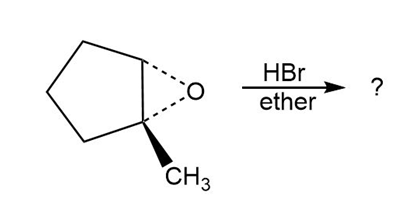
c.
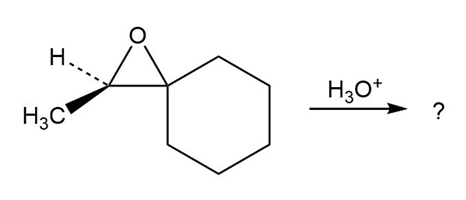
d.
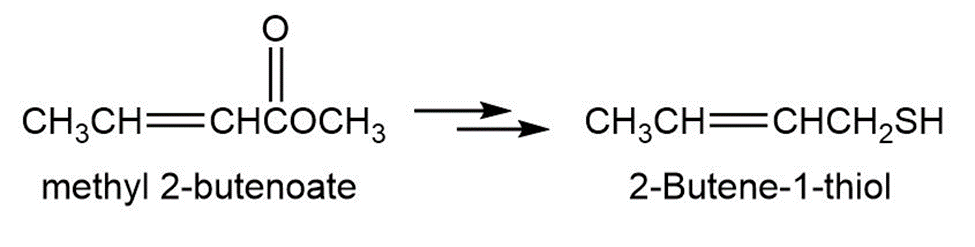
2-Butene-1-thiol is one component of skunk spray. How would you synthesize this substance from methyl 2-butenoate? From 1,3-butadiene?
Predict the products of the following reactions:
(a)
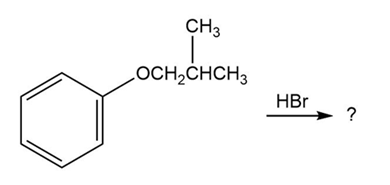
(b)
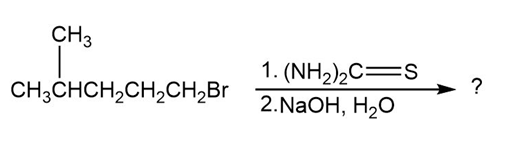
(c)
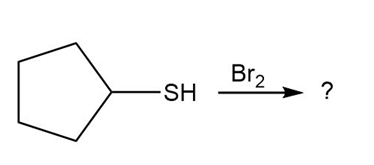
(d)
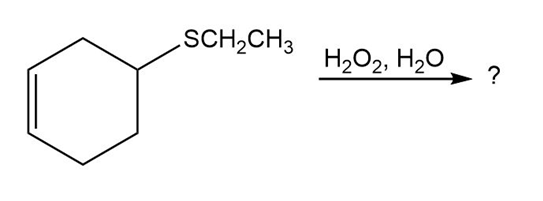
Rank the following halides in order of their reactivity in Williamson synthesis:
Imagine that you have created (2R, 3R)-2,3-epoxy-3-methylpentane with aqueous acid to carry out a ring opening reaction.
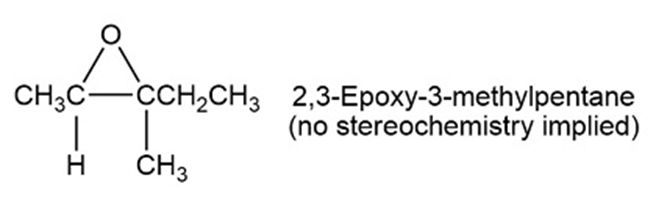
What do you think about this solution?
We value your feedback to improve our textbook solutions.