Chapter 14: Q 14-14-34 E (page 447)
How can you account for the fact that cis-1,3-pentadiene is much less reactive than trans-1,3-pentadiene in Diels-Alder reaction.
Short Answer
Due to steric reasons.
Learning Materials
Features
Discover
Chapter 14: Q 14-14-34 E (page 447)
How can you account for the fact that cis-1,3-pentadiene is much less reactive than trans-1,3-pentadiene in Diels-Alder reaction.
Due to steric reasons.
All the tools & learning materials you need for study success - in one app.
Get started for free
Give IUPAC names for the following compounds:
(a)
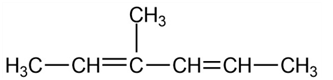
Predict the products of the following Diels-Alder reaction:
(a) In the light of your answer to problem to propose mechanism for the reactions below.
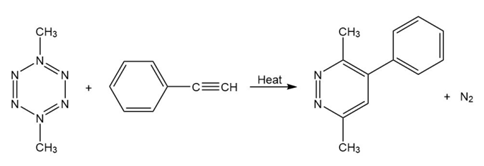
Question: 1,3,5-Hexatriene has =258 nm. In light of your answer to Problem
14-48, approximately where would you expect 2,3-dimethyl-1,3,5-hexatriene to absorb?
Calculate the energy range of electromagnetic radiation in the UV region of the spectrum from 200 to 400 nm (see Section 12-5). How does this value compare with the values calculated previously for IR and NMR spectroscopy?
What do you think about this solution?
We value your feedback to improve our textbook solutions.