Chapter 14: 25Ea (page 447)
Give IUPAC names for the following compounds:
(a)
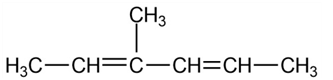
Short Answer
3-Methyl-2,4-hexa-diene
Learning Materials
Features
Discover
Chapter 14: 25Ea (page 447)
Give IUPAC names for the following compounds:
(a)

3-Methyl-2,4-hexa-diene
All the tools & learning materials you need for study success - in one app.
Get started for free
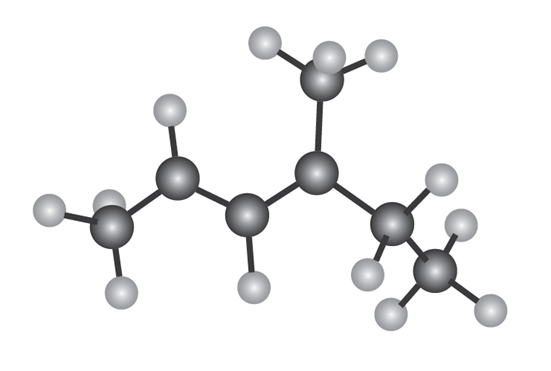
The following model is that of an allylic carbocation intermediateformed by protonation of a conjugated diene with HBr. Show the structureof the diene and the structures of the final reaction products.
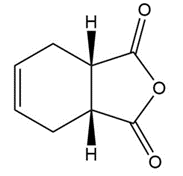
How could you use Diels-Alder reactions to prepare the following products ? Show the starting diene and dienophile in each case.
a)
1,3-Cyclopentadiene polymerizes slowly at room temperature to yield
a polymer that has no double bonds except on the ends. On heating, the
polymer breaks down to regenerate 1,3-cyclopentadiene. Propose a structure for the product.
The following ultraviolet absorption maxima have been measured:
1,3-Butadiene 217 nm
2-Methyl-1,3-butadiene 220 nm
1,3-Pentadiene 223 nm
2,3-Dimethyl-1,3-butadiene 226 nm
2,4-Hexadiene227 nm
2,4-Dimethyl-1,3-pentadiene 232 nm
2,5-Dimethyl-2,4-hexadiene 240 nm
What conclusion can you draw about the effect of alkyl substitution on
UV absorption maxima? Approximately what effect does each added
alkyl group have?
Dimethyl butynedioate undergoes a Diels-Alder reaction with (2E,4E)-2,4-hexadiene. Show the structure and stereochemistry of the product.
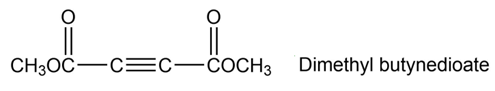
What do you think about this solution?
We value your feedback to improve our textbook solutions.