Chapter 14: 32E (page 447)
Di-tert-butyl-1,3-butadiene does not undergo Diels-Alder reactions. Explain.
Short Answer
Due to steric hinderance.
Learning Materials
Features
Discover
Chapter 14: 32E (page 447)
Di-tert-butyl-1,3-butadiene does not undergo Diels-Alder reactions. Explain.
Due to steric hinderance.
All the tools & learning materials you need for study success - in one app.
Get started for free
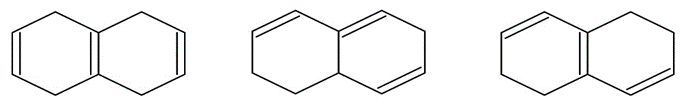
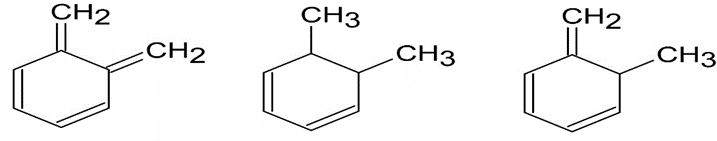
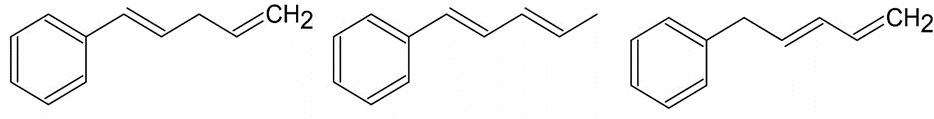
Arrange the molecules according to where you would expect to find
their wavelength of maximum absorption in UV spectroscopy, from shortest to longest wavelength.
a)
b)
c)
How could you use Diels-Alder reactions to prepare the following products ? Show the starting diene and dienophile in each case.
c)
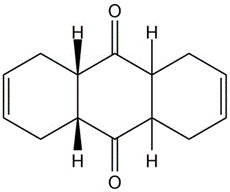
The following model is that of an allylic carbocation intermediateformed by protonation of a conjugated diene with HBr. Show the structureof the diene and the structures of the final reaction products.
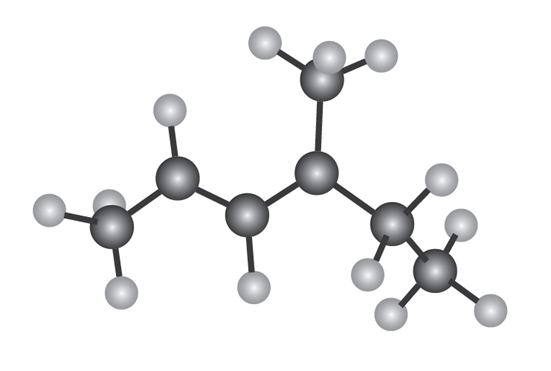
Electrophilic addition of to isoprene (2-methyl-1,3-butadiene) yields the following product mixture:
Of the 1,2-addition products, explain why 3,4-dibromo-3-methyl-1-butene (21%) predominates over 3,4-dibromo-2-methyl-1-butene (3%).
Why do you suppose 1,4 adducts of 1,3-butadiene are generally more stable than 1,2 adducts?
What do you think about this solution?
We value your feedback to improve our textbook solutions.