Chapter 14: 30E (page 447)
Draw the possible products resulting from addition of 1 equivalent of HCl to 1-phenyl-1,3-buta-di-ene. Which would you expect to predominate, and why?
Short Answer
2-chloro-1-phenyl- butene will predominate.
Learning Materials
Features
Discover
Chapter 14: 30E (page 447)
Draw the possible products resulting from addition of 1 equivalent of HCl to 1-phenyl-1,3-buta-di-ene. Which would you expect to predominate, and why?
2-chloro-1-phenyl- butene will predominate.
All the tools & learning materials you need for study success - in one app.
Get started for free
Would you expect allene, , to show a UV absorption in
the 200 to 400 nm range? Explain.
Calculate the energy range of electromagnetic radiation in the UV region of the spectrum from 200 to 400 nm (see Section 12-5). How does this value compare with the values calculated previously for IR and NMR spectroscopy?
Question:Ocimene is a pleasant-smelling hydrocarbon found in the leaves of
certain herbs. It has the molecular formula and a UV absorption
maximum at 232 nm. On hydrogenation with a palladium catalyst,
2,6-dimethyloctane is obtained. Ozonolysis of Ocimene, followed by
treatment with zinc and acetic acid, produces the following four
fragments:
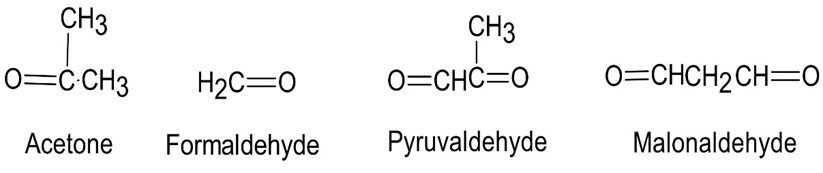
(a)How many double bonds does ocimene have?
(b)Is ocimene conjugated or non-conjugated?
(c)Propose a structure for ocimene.
(d)Write the reactions, showing starting material and products.
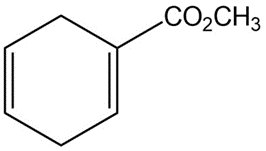
How could you use Diels-Alder reactions to prepare the following products ? Show the starting diene and dienophile in each case.
a)
1,3-Cyclopentadiene polymerizes slowly at room temperature to yield
a polymer that has no double bonds except on the ends. On heating, the
polymer breaks down to regenerate 1,3-cyclopentadiene. Propose a structure for the product.
What do you think about this solution?
We value your feedback to improve our textbook solutions.