Chapter 14: 29E (page 447)
Propose a structure for a conjugated diene that gives the same product from both 1,2 and 1,4-addition of HBr.
Short Answer
Cyclopentadiene
Learning Materials
Features
Discover
Chapter 14: 29E (page 447)
Propose a structure for a conjugated diene that gives the same product from both 1,2 and 1,4-addition of HBr.
Cyclopentadiene
All the tools & learning materials you need for study success - in one app.
Get started for free
Dimethyl butynedioate undergoes a Diels-Alder reaction with (2E,4E)-2,4-hexadiene. Show the structure and stereochemistry of the product.
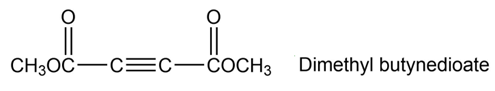
Question: In the light of your answer to problem to propose mechanism for the reactions below.
Norboradiene (Problem 14-55) can be prepared by reaction of chloroethylene with 1,3-cyclopentadiene, followed by treatment of the product with sodium ethoxide. Write the overall scheme and identify the two kinds of reactions.
Give IUPAC names for the following compounds:
How could you use Diels-Alder reactions to prepare the following products ? Show the starting diene and dienophile in each case.
c)
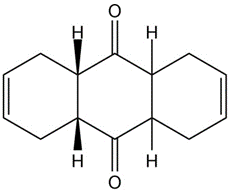
What do you think about this solution?
We value your feedback to improve our textbook solutions.