Chapter 14: 27Ea (page 447)
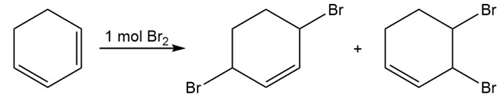
What product (s) would you expect to obtain from reaction of 1,3-cyclohexadiene with each of the following?
(a) 1 mol in
Short Answer
Learning Materials
Features
Discover
Chapter 14: 27Ea (page 447)
What product (s) would you expect to obtain from reaction of 1,3-cyclohexadiene with each of the following?
(a) 1 mol in

All the tools & learning materials you need for study success - in one app.
Get started for free
Why do you suppose 1,4 adducts of 1,3-butadiene are generally more stable than 1,2 adducts?
Answer the questions below for 1,3,5-cycloheptatriene.
(a)How many patomic orbitals are in the conjugated system?
(b)How many molecular orbitals describe the conjugated system?
(c)How many molecular orbitals are bonding molecular orbitals?
(d)How many molecular orbitals are anti-bonding molecular orbitals?
(e)Which molecular orbitals are filled with electrons?
(f)If this molecule were to absorb a photon of UV light an electron would move between which two molecular orbitals (be specific)?
How could you use Diels-Alder reactions to prepare the following products ? Show the starting diene and dienophile in each case.
a)

Predict the products of the following Diels-Alder reaction:
(b)
Draw the possible products resulting from addition of 1 equivalent of HCl to 1-phenyl-1,3-buta-di-ene. Which would you expect to predominate, and why?
What do you think about this solution?
We value your feedback to improve our textbook solutions.