Chapter 16: Q26E (page 524)
How would you synthesize the following compound starting from benzene? More than one step is needed.
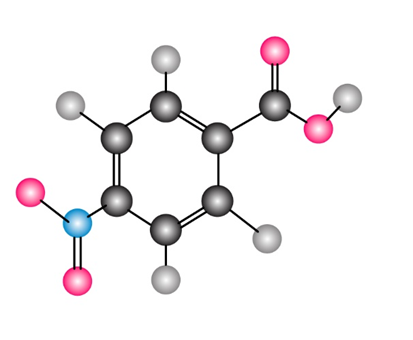
Short Answer
Learning Materials
Features
Discover
Chapter 16: Q26E (page 524)
How would you synthesize the following compound starting from benzene? More than one step is needed.


All the tools & learning materials you need for study success - in one app.
Get started for free
Draw resonance structures for the intermediates from the reaction of an electrophile at the ortho, meta, and para positions of nitrobenzene. Which intermediates are most stable?
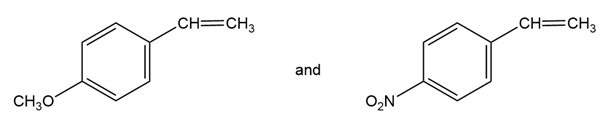
You know the mechanism of HBr addition to alkenes, and you know the effects of various substituent groups on aromatic substitution. Use this knowledge to predict which of the following two alkenes reacts faster with HBr. Explain your answer by drawing resonance structures of the carbocation intermediates.
The N,N,N-trimethylammonium group , is one of the few groups that is a meta-directing deactivator yet has no electron-withdrawing resonance effect. Explain
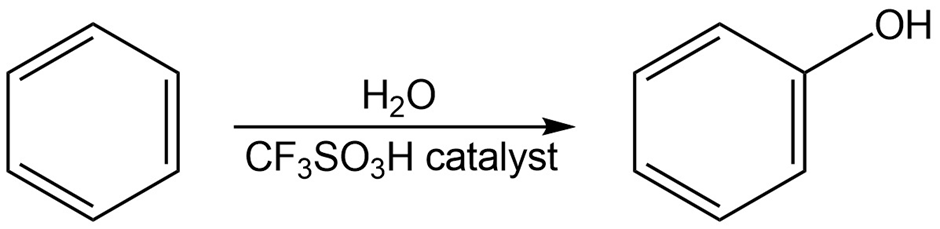
Benzene and alkyl-substituted benzenes can be hydroxylated by reaction with in the presence of an acidic catalyst. What is the structure of the reactive electrophile? Propose a mechanism for the reaction.
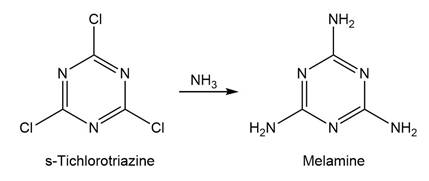
Question:Melamine, used as a fire retardant and a component of the writing surface of white boards, can be prepared from s-trichlorotriazine through a series of SNAr reactions with ammonia. The first substitution takes place rapidly at room temperature. The second substitution takes place near 100 °C, and the third substitution requires even higher temperature and pressure. Provide an explanation for this reactivity.
What do you think about this solution?
We value your feedback to improve our textbook solutions.