Chapter 20: Q38P (page 678)
Calculate the pKa's for the following acids:
(a) Lactic acid, Ka = 8.4 × 10-4
(b) Acrylic acid, Ka = 5.6 × 10-6
Short Answer
Thevalue of pKa for lactic and acrylic acid were calculated.
Learning Materials
Features
Discover
Chapter 20: Q38P (page 678)
Calculate the pKa's for the following acids:
(a) Lactic acid, Ka = 8.4 × 10-4
(b) Acrylic acid, Ka = 5.6 × 10-6
Thevalue of pKa for lactic and acrylic acid were calculated.
All the tools & learning materials you need for study success - in one app.
Get started for free
20-36 Order the compounds in each of the following sets with respect to increasing acidity:
(a) Acetic acid, oxalic acid, formic acid
(b) p-Bromobenzoic acid, p-nitrobenzoic acid, 2,4-dinitrobenzoic acid
(c) Fluoroacetic acid, 3-fluoropropanoic acid, iodoacetic acid
Which method – Grignard carboxylation or nitrile hydrolysis would you use for each of the following reactions? Explain.
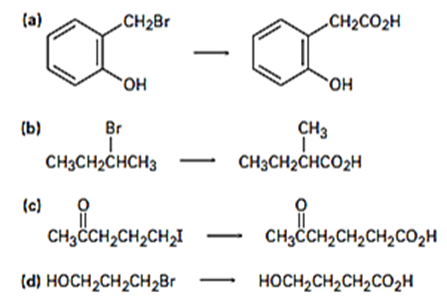
Predict the product (s) and provide the mechanism for each reaction below.
Propose a structure for a compound that dissolves in dilute NaOH and shows the following 1H NMR spectrum: 1.08 (9H, singlet), 2.2 (2H, singlet), and 11.2 (1H, singlet).
Give IUPAC names for the following compounds:
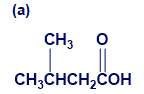
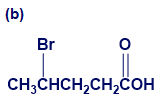
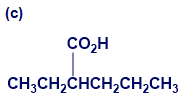
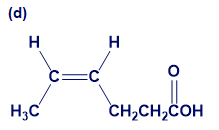
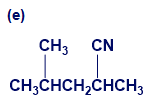
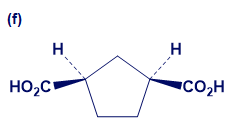
What do you think about this solution?
We value your feedback to improve our textbook solutions.