Chapter 20: Q22E (page 678)
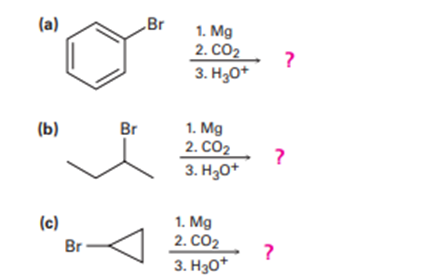
Predict the product (s) and provide the mechanism for each reaction below.
Short Answer
The product prediction and its mechanism for each of the reaction can be explained.
Learning Materials
Features
Discover
Chapter 20: Q22E (page 678)
Predict the product (s) and provide the mechanism for each reaction below.
The product prediction and its mechanism for each of the reaction can be explained.
All the tools & learning materials you need for study success - in one app.
Get started for free
Predict the product and provide the mechanism for each reaction below.
The of p-cyclopropylbenzoic acid is 4.45. Is cyclopropylbenzene likely to be more reactive or less reactive than benzene toward electrophilic bromination? Explain.
Draw structures corresponding to the following IUPAC names:
Hepatanedioic acid
2-Hexen-4-ynoic acid
4-Ethyl-2-propyloctanoic acid
3-chlorophthalic acid
Triphenylacetic acid
2-Cyclobutenecarbonitrile
m-Benzoyl benzonitrile
Pregabalin, marketed as Lyrica, is an anticonvulsant drug that is also effective in treating chronic pain. The IUPAC name of pregabalin is (S)-3-(aminomethyl)-5-methyl hexanoic acid. (An aminomethyl group is - .) Draw the structure of pregabalin.
Acid-catalyzed hydrolysis of a nitrile to give a carboxylic acid occurs by initial protonation of the nitrogen atom, followed by nucleophilic addition of water. Review the mechanism of base-catalyzed nitrile hydrolysis in Section 20-7 and then predict the products for each reaction below and write all of the steps involved in the acid-catalyzed reaction, using curved arrows to represent electron flow in each step.
What do you think about this solution?
We value your feedback to improve our textbook solutions.