Chapter 20: Q30E (page 678)
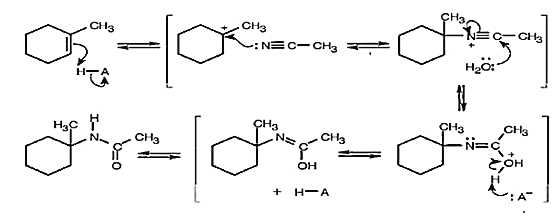
In the Ritter reaction, an alkene reacts with a nitrile in the presence of strong aqueous sulfuric acid to yield an amide. Propose a mechanism.
Short Answer
A mechanism of Ritter reaction is shown below
Learning Materials
Features
Discover
Chapter 20: Q30E (page 678)
In the Ritter reaction, an alkene reacts with a nitrile in the presence of strong aqueous sulfuric acid to yield an amide. Propose a mechanism.
A mechanism of Ritter reaction is shown below

All the tools & learning materials you need for study success - in one app.
Get started for free
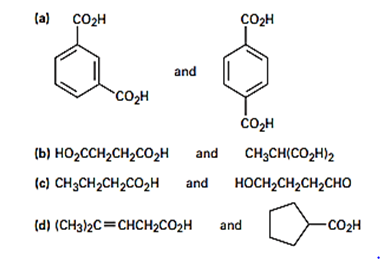
How would you use NMR to distinguish between the following pairs of isomers?
Arrange the compounds in each of the following sets in order of increasing basicity:
(a) Magnesium acetate, magnesium hydroxide, methyl magnesium bromide
(b) Sodium benzoate, sodium p-nitrobenzoate, sodium acetylide
(c) Lithium hydroxide, lithium ethoxide, lithium formate
1,6 Hexanediamine, a starting material needed for making nylon, can be made from 1,3 butadiene. How would you accomplish the synthesis?
How could you convert butanoic acid into the following compounds?
Write each step showing the reagents needed.
(a) 1-Butanol
(b) 1-Bromobutane
(c) Pentanoic acid
(d) 1-Butene
(e) Octane
Cyclopentanecarboxylic acid and 4-hydroxycyclohexanone have the
same formula , and both contain an -OH and a C=O group. How could
you distinguish between them using IR spectroscopy?
What do you think about this solution?
We value your feedback to improve our textbook solutions.