Chapter 20: Q21E (page 678)
Predict the product and provide the mechanism for each reaction below.
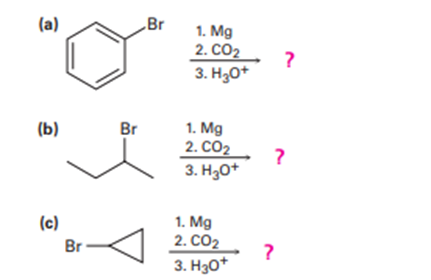
Short Answer
The product prediction and its mechanism for each of the reaction can be explained.
Learning Materials
Features
Discover
Chapter 20: Q21E (page 678)
Predict the product and provide the mechanism for each reaction below.

The product prediction and its mechanism for each of the reaction can be explained.
All the tools & learning materials you need for study success - in one app.
Get started for free
The two 1H NMR spectrashown here belong to crotonic acid (trans-) and methacrylic acid (). Which spectrum corresponds to which acid? Explain.
How would you prepare 1-phenyl-2-butanone, , from benzyl bromide, ? More than one step is required.
Isocitric acid, an intermediate in the citric acid cycle of food metabolism, has the systematic name (2R,3S)-3-carboxy-2-hydroxypentane-dioic acid. Draw the structure.
1,6 Hexanediamine, a starting material needed for making nylon, can be made from 1,3 butadiene. How would you accomplish the synthesis?
In humans, the final product of purine degradation from DNA is uric acid, pKa = 5.61, which is excreted in the urine. What is the percent dissociation of uric acid in urine at a typical pH = 6.0? Why do you think uric acid is acidic even though it does not have a CO₂H group?
What do you think about this solution?
We value your feedback to improve our textbook solutions.