Chapter 20: Carboxylic Acids and Nitriles
Q36P
20-36 Order the compounds in each of the following sets with respect to increasing acidity:
(a) Acetic acid, oxalic acid, formic acid
(b) p-Bromobenzoic acid, p-nitrobenzoic acid, 2,4-dinitrobenzoic acid
(c) Fluoroacetic acid, 3-fluoropropanoic acid, iodoacetic acid
Q37P
Arrange the compounds in each of the following sets in order of increasing basicity:
(a) Magnesium acetate, magnesium hydroxide, methyl magnesium bromide
(b) Sodium benzoate, sodium p-nitrobenzoate, sodium acetylide
(c) Lithium hydroxide, lithium ethoxide, lithium formate
Q38P
Calculate the pKa's for the following acids:
(a) Lactic acid, Ka = 8.4 × 10-4
(b) Acrylic acid, Ka = 5.6 × 10-6
Q39P
Calculate the Ka's for the following acids:
(a) Citric acid, pKa = 3.14
(b) Tartaric acid, pKa = 2.98
Q3P
Assume you have a mixture of naphthalene and benzoic acid that you want to separate. How might you take advantage of the acidity of one component in the mixture to effect a separation?
Q40E
Thioglycolic acid, HSCH₂CO₂H, a substance used in depilatory agents(hair removers) has pKa = 3.42. What is the percent dissociation ofthioglycolic acid in a buffer solution at pH = 3.0?
Q40P
Thioglycolic acid, , a substance used in depilatory agents
(hair removers) has. What is the percent dissociation of
thioglycolic acid in a buffer solution at ?
Q41E
In humans, the final product of purine degradation from DNA is uricacid, pKa = 5.61, which is excreted in the urine. What is the percent dissociation of uric acid in urine at a typical pH = 6.0? Why do you think uric acid is acidic even though it does not have a CO₂H group?
Q41P
In humans, the final product of purine degradation from DNA is uric acid, , which is excreted in the urine. What is the percentdissociation of uric acid in urine at a typical ? Why do you think uric acid is acidic even though it does not have a group?
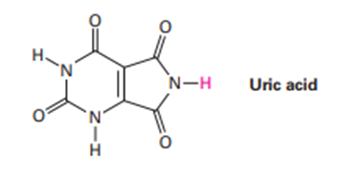
Q42E
In humans, the final product of purine degradation from DNA is uric acid, pKa = 5.61, which is excreted in the urine. What is the percent dissociation of uric acid in urine at a typical pH = 6.0? Why do you think uric acid is acidic even though it does not have a CO₂H group?


