Chapter 23: Q9P (page 764)
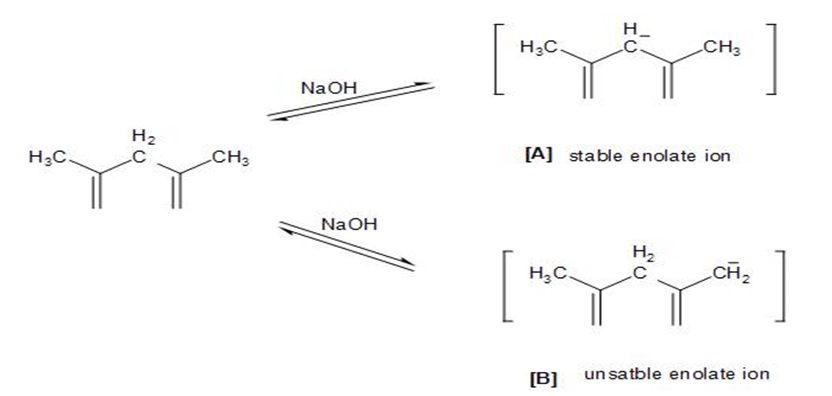
Treatment of a 1,3-diketone such as 2,4-pentanedione with base does not give an aldol condensation product. Explain.
Short Answer
Due to the formation of the stable compound at equilibrium, treatment of a 1,3-diketone such as 2,4-pentanedione with base does not give an aldol condensation product.