Chapter 23: Q28E (page 783)
Based on your answers to Problem 23-27, predict the dehydration product for each reaction and provide the mechanism.
Short Answer
The products are formed by dehydration reaction.
Learning Materials
Features
Discover
Chapter 23: Q28E (page 783)
Based on your answers to Problem 23-27, predict the dehydration product for each reaction and provide the mechanism.
The products are formed by dehydration reaction.
All the tools & learning materials you need for study success - in one app.
Get started for free
How could you prepare the following cyclohexanones by combining a Stork enamine reaction with an intramolecular aldol condensation? (See Problem 23-64.)
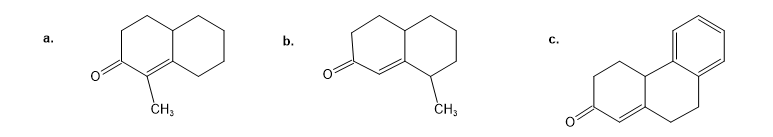
Which of the following compounds would you expect to undergo aldol self-condensation? Show the product of each successful reaction.
(a) Trimethyl acetaldehyde
(b) Cyclobutanone
(c) Benzophenone (diphenyl ketone)
(d) 3-Pentanone
(e) Decanal
(f) 3-Phenyl-2-propenal
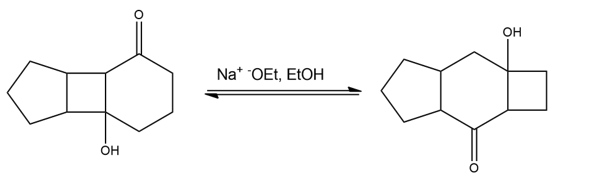
Propose a mechanism for the following base-catalyzed isomerization:
Dieckmann cyclization of diethyl 3-methylheptanedioate gives a mixture of two𝛃-keto ester products. What are their structures, and why is a mixture formed?
Which of the following compounds can probably be prepared by a mixed aldol reaction? Show the reactants you would use in each case.
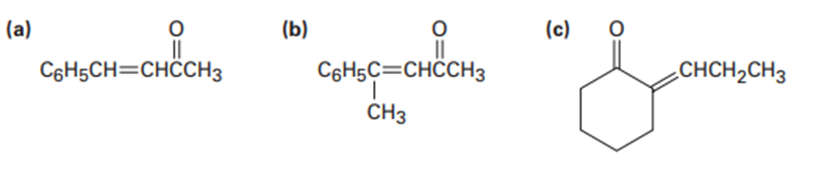
What do you think about this solution?
We value your feedback to improve our textbook solutions.