Chapter 23: Q44E (page 783)
Propose a mechanism for the following base-catalyzed isomerization:
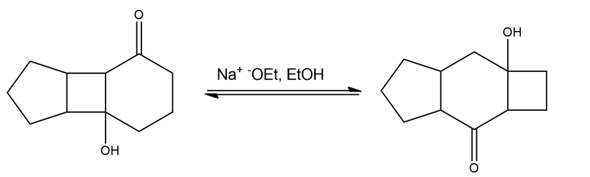
Short Answer
The product is formed by the base-catalyzed isomerization.
Learning Materials
Features
Discover
Chapter 23: Q44E (page 783)
Propose a mechanism for the following base-catalyzed isomerization:

The product is formed by the base-catalyzed isomerization.
All the tools & learning materials you need for study success - in one app.
Get started for free
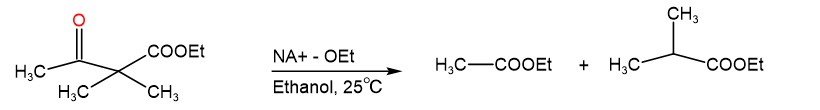
Ethyl dimethyl acetoacetate reacts instantly at room temperature when treated with ethoxide ion to yield two products, ethyl acetate, and ethyl 2-methyl propanoate. Propose a mechanism for this cleavage reaction.
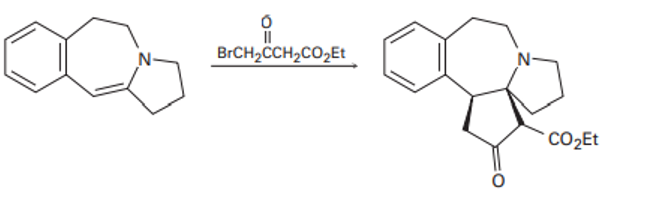
Propose a mechanism to account for the following reaction of an enamine with an alkyl halide:
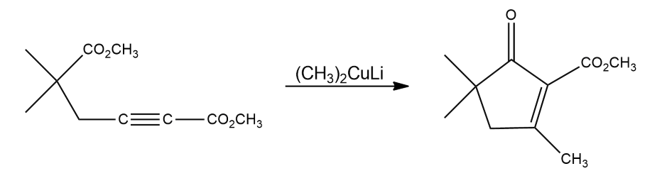
The following reaction involves a conjugate addition reaction followed by an intramolecular Claisen condensation. Write both steps, and show their mechanisms.
The following reaction involves two successive intramolecular Michael reactions. Write both steps, and show their mechanisms.
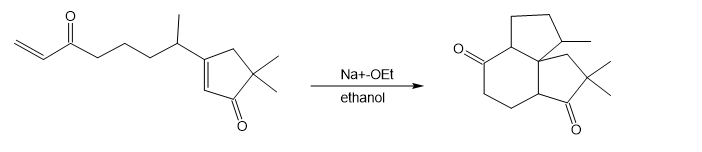
The following reaction involves an intramolecular Michael reaction followed by an intramolecular aldol reaction. Write both steps, and show their mechanisms.
What do you think about this solution?
We value your feedback to improve our textbook solutions.