Chapter 23: Q47E (page 783)
Propose a mechanism to account for the following reaction of an enamine with an alkyl halide:
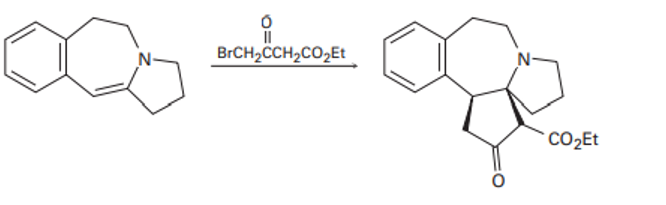
Short Answer
The above reaction is the Stork Enamine Alkylation reaction.
Learning Materials
Features
Discover
Chapter 23: Q47E (page 783)
Propose a mechanism to account for the following reaction of an enamine with an alkyl halide:

The above reaction is the Stork Enamine Alkylation reaction.
All the tools & learning materials you need for study success - in one app.
Get started for free
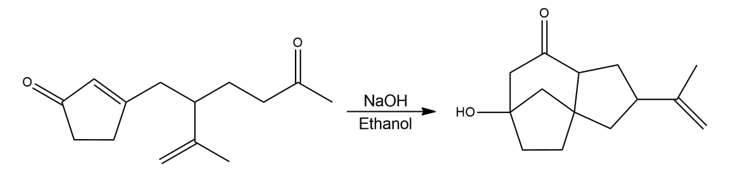
The following reaction involves an intramolecular Michael reaction followed by an intramolecular aldol reaction. Write both steps, and show their mechanisms.
The major product formed by intramolecular aldol cyclization of 2,5-heptanedione (Problem 23-51) has two singlet absorptions in the 1H NMR spectrum, at 1.65 d and 1.90 d, and has no absorptions in the range 3 to 10 d. What is its structure?
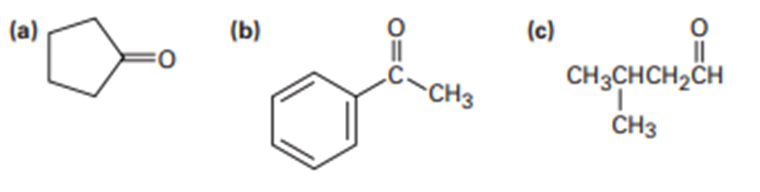
What enone product would you expect from aldol condensation of each of the following compounds?
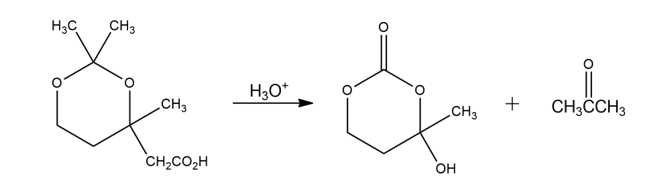
The following reaction involves a hydrolysis followed by an intramolecular nucleophilic acyl substitution reaction. Write both steps, and show their mechanisms.
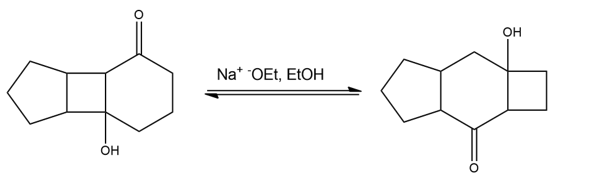
Propose a mechanism for the following base-catalyzed isomerization:
What do you think about this solution?
We value your feedback to improve our textbook solutions.