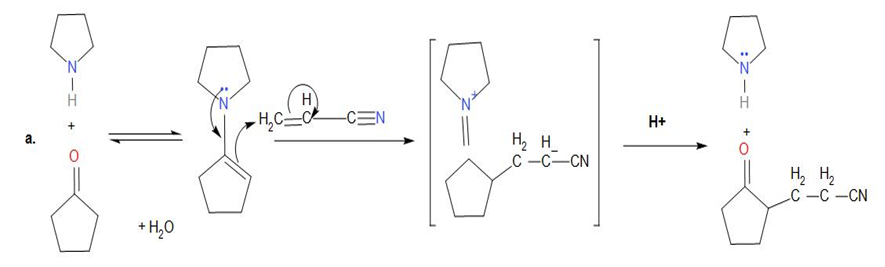
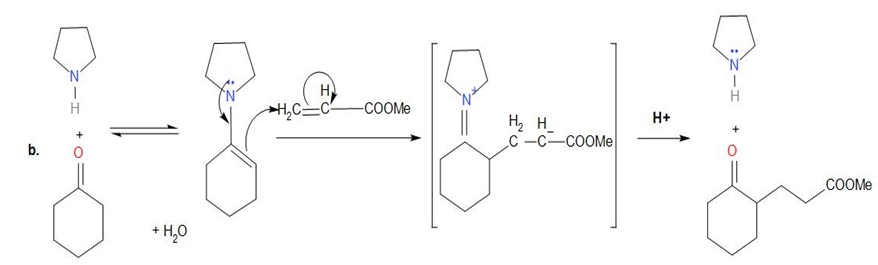
Chapter 23: Q23-20P (page 775)
Show how you might use an enamine reaction to prepare each of the following compounds:
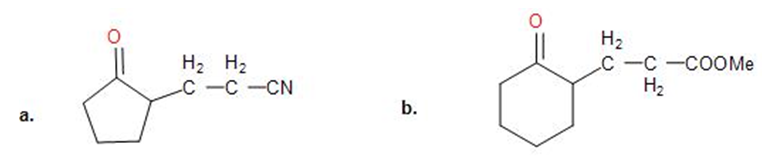
Short Answer
Learning Materials
Features
Discover
Chapter 23: Q23-20P (page 775)
Show how you might use an enamine reaction to prepare each of the following compounds:
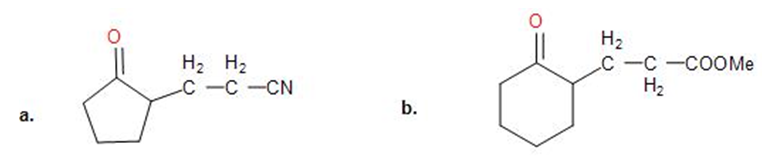


All the tools & learning materials you need for study success - in one app.
Get started for free
Azlactones are important starting materials used in the synthesis of dehydro a-aminoacids. They react with aldehydes to form an intermediate that is hydrolyzed under acidic conditions to give the final amino acid product. Provide the structure of the intermediate and propose a mechanism for its formation.
How might the following compounds be prepared using Michael reactions? Show the nucleophilic donor and the electrophilic acceptor in each case.
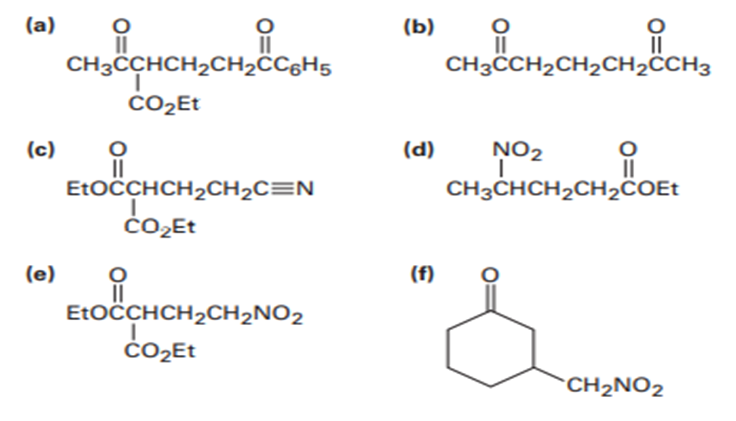
The Knoevenagel reaction is a carbonyl condensation reaction of an ester with an aldehyde or ketone to yield an α,β-unsaturated product. Show the mechanism for the Knoevenagel reaction of diethyl malonate with benzaldehyde.
Dieckmann cyclization of diethyl 3-methylheptanedioate gives a mixture of two𝛃-keto ester products. What are their structures, and why is a mixture formed?
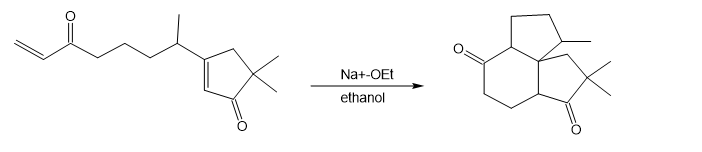
The following reaction involves two successive intramolecular Michael reactions. Write both steps, and show their mechanisms.
What do you think about this solution?
We value your feedback to improve our textbook solutions.