Chapter 23: Q18P (page 773)
How would you prepare the following compound using a Michael reaction?
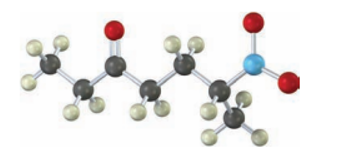
Short Answer
By the reaction between pent-1-ene-3-one and nitroethane we can prepare the given compound.
Learning Materials
Features
Discover
Chapter 23: Q18P (page 773)
How would you prepare the following compound using a Michael reaction?

By the reaction between pent-1-ene-3-one and nitroethane we can prepare the given compound.
All the tools & learning materials you need for study success - in one app.
Get started for free
Isoleucine, another of the twenty amino acids found in proteins, metabolized by a pathway that includes the following step. Propose a mechanism.
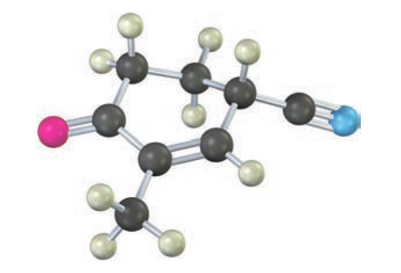
The following molecule was formed by a Robinson annulation reaction. What reactants were used?
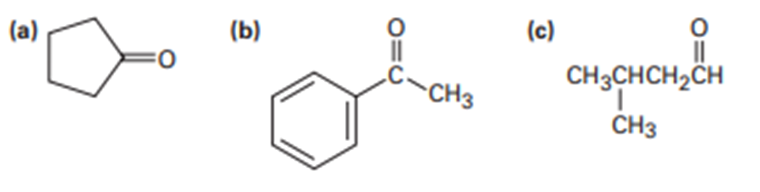
Aldol condensation of 3-methyl cyclohexanone leads to a mixture of two enone products, not counting double-bond isomers. Draw them.
The major product formed by intramolecular aldol cyclization of 2,5-heptanedione (Problem 23-51) has two singlet absorptions in the 1H NMR spectrum, at 1.65 d and 1.90 d, and has no absorptions in the range 3 to 10 d. What is its structure?
What enone product would you expect from aldol condensation of each of the following compounds?
What do you think about this solution?
We value your feedback to improve our textbook solutions.