Chapter 23: Q14P (page 770)
What product would you expect from the following reaction?
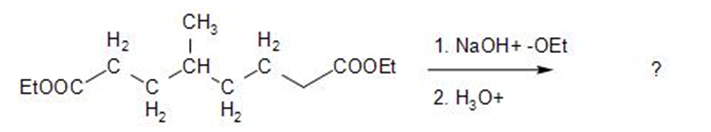
Short Answer
The product is formed when diethyl 4-methyl heptane dioate is
Ethyl 4-methyl 2-oxocyclohexanecarboxylate.
Learning Materials
Features
Discover
Chapter 23: Q14P (page 770)
What product would you expect from the following reaction?

The product is formed when diethyl 4-methyl heptane dioate is
Ethyl 4-methyl 2-oxocyclohexanecarboxylate.
All the tools & learning materials you need for study success - in one app.
Get started for free
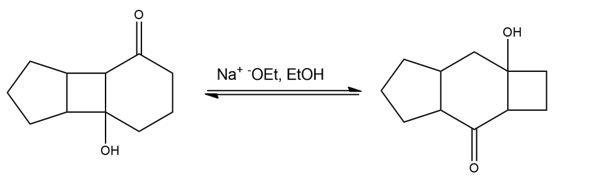
Propose a mechanism for the following base-catalyzed isomerization:
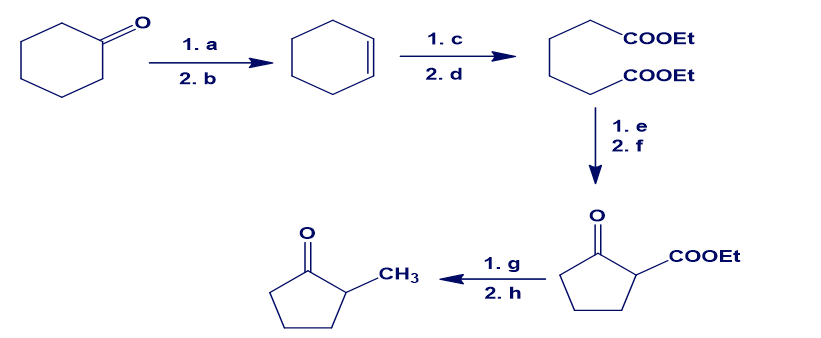
Fill in the missing reagents a–h in the following scheme:
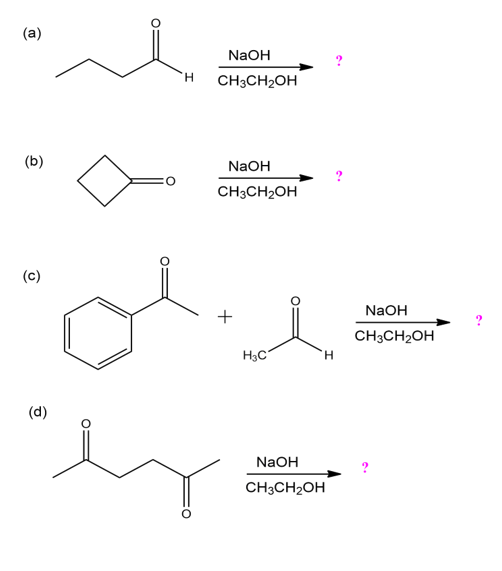
Predict the addition product for each reaction below and provide the mechanism.
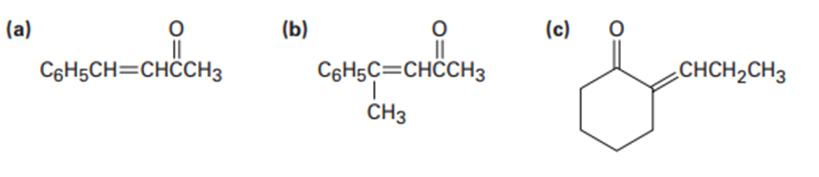
Which of the following compounds can probably be prepared by a mixed aldol reaction? Show the reactants you would use in each case.
In the mixed Claisen reaction of cyclopentanone with ethyl format, a much higher yield of the desired product is obtained by first mixing the two carbonyl components and then adding a base, rather than by first mixing base with cyclopentanone and then adding ethyl format. Explain.
What do you think about this solution?
We value your feedback to improve our textbook solutions.