Chapter 23: Q12P (page 766)
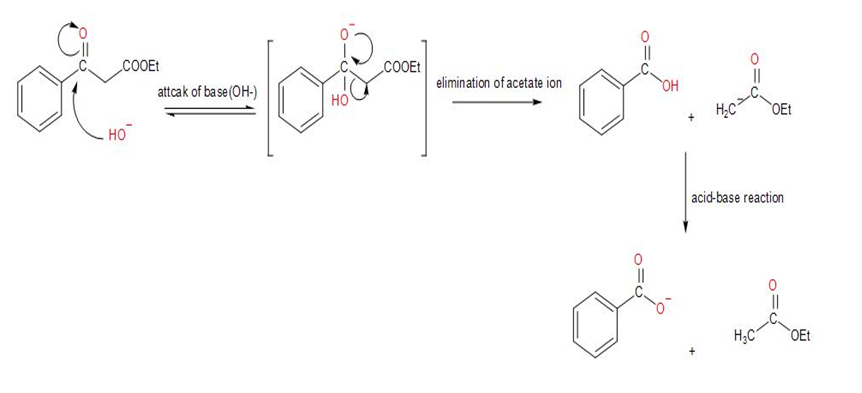
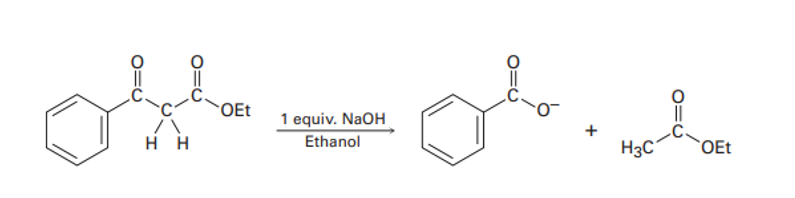
As shown in Figures 23-4, the Claisen reaction is reversible. That is, a𝛃-keto ester can be cleaved by base into two fragments. Using curved arrows to indicate electron flow, show the mechanism by which this cleavage occurs.
Short Answer
When a 𝛃-keto ester reacts with a strong base (NaOH) it can give us two products but one of them is reversible and can’t give the desired product.