Chapter 6: 6-33a (page 181)
For each reaction below identify the electrophile and the nucleophile
a.
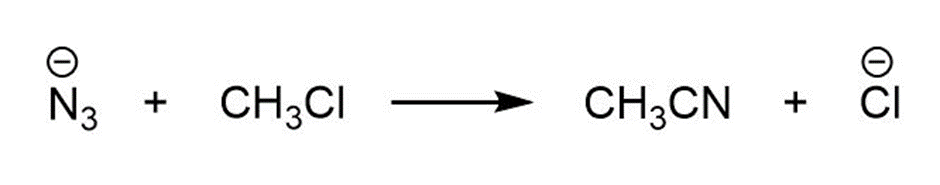
b.
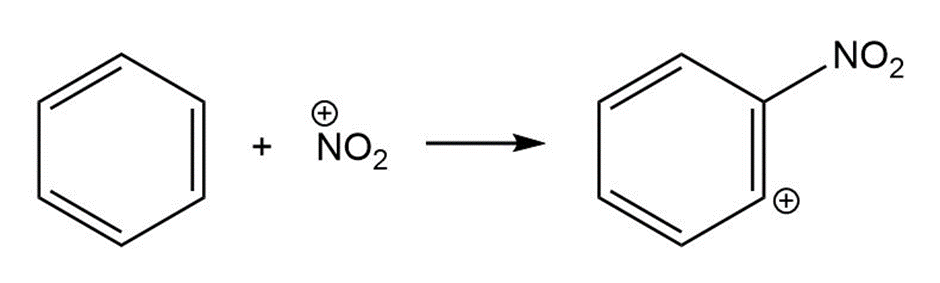
c.
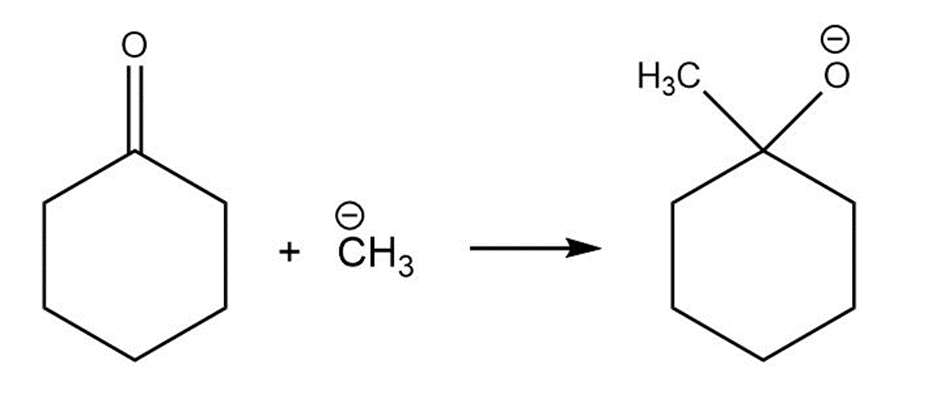
Short Answer
a.
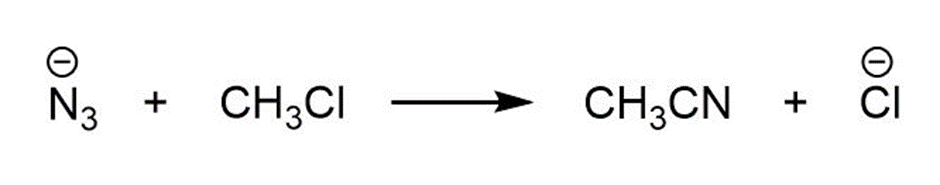
Learning Materials
Features
Discover
Chapter 6: 6-33a (page 181)
For each reaction below identify the electrophile and the nucleophile
a.

b.

c.

a.

All the tools & learning materials you need for study success - in one app.
Get started for free
Identify the likely electrophilic and nucleophilic sites in each of the following molecules:
a.
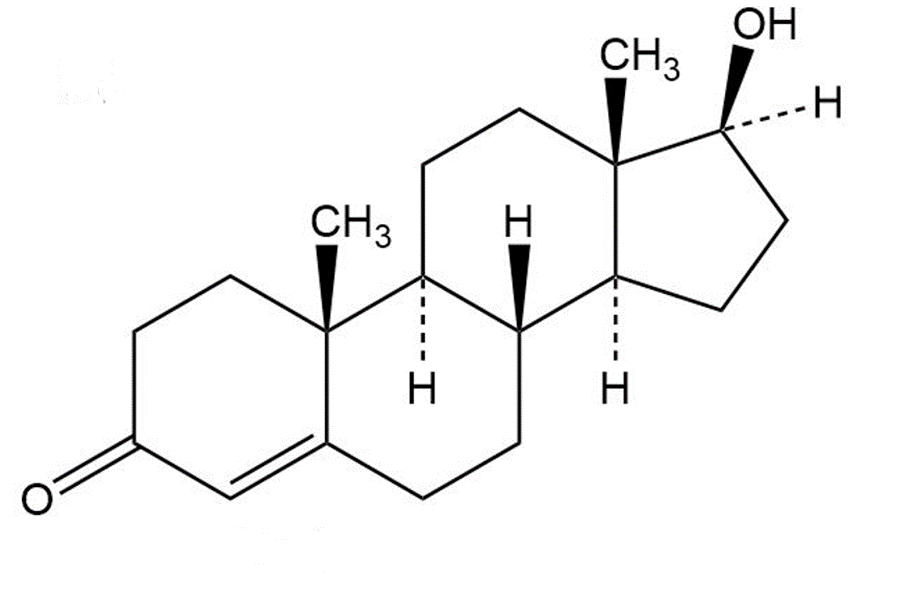
testosterone
b.
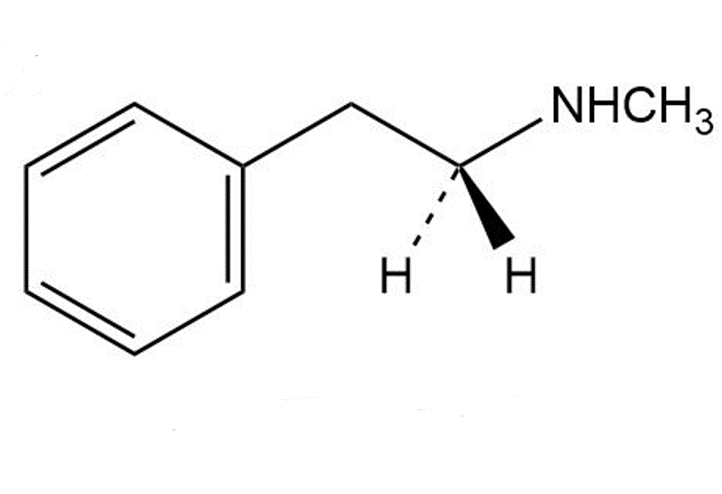
Methanphetamine
When a mixture of methane and chlorine is irradiated, a reaction commences immediately. When irradiation is stopped, the reaction gradually slows down but does not stop immediately. Explain.
Despite the limitations of radical chlorination of alkanes, the reaction is still useful for synthesizing certain halogenated compounds. For which of the following compounds does radical chlorination give a single monochloro product?
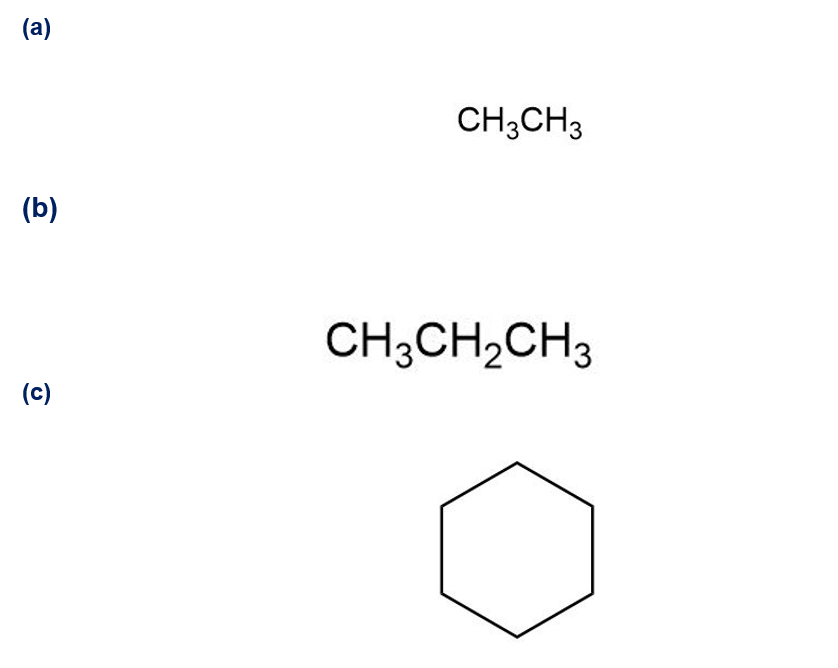
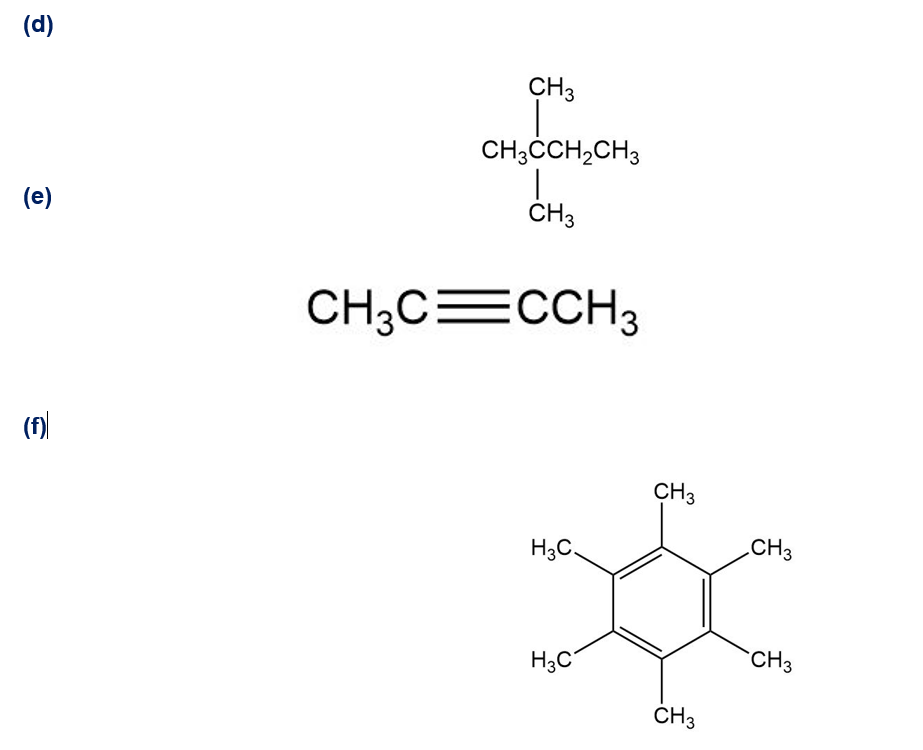
Follow the flow of electrons indicated by the curved arrows in each of the following polar reactions, and predict the products that result:
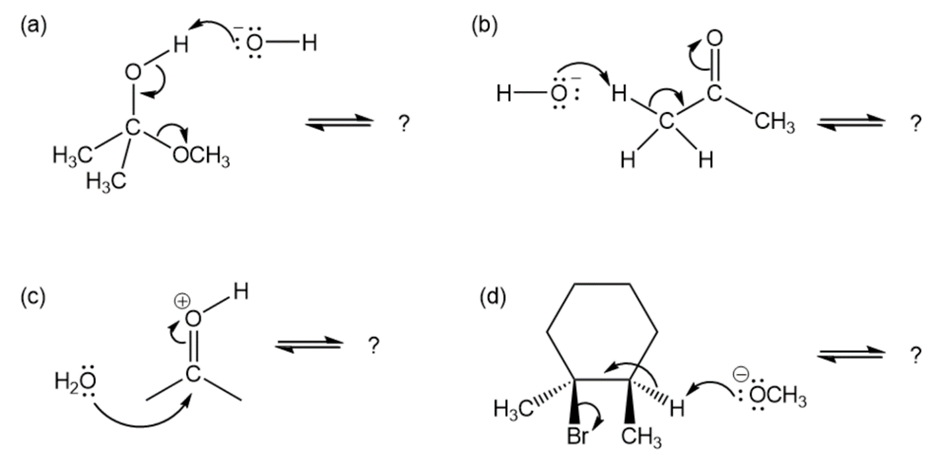
Which reaction is likely to be more exergonic, one with =1000 or one with = 0.001?
What do you think about this solution?
We value your feedback to improve our textbook solutions.